CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles. Walk-in opportunities for multiple experience levels.
CEPH Lifesciences has announced multiple job openings across its pharmaceutical manufacturing facility in Baddi, Himachal Pradesh. The company is expanding its workforce and inviting applications from qualified professionals across production, quality, microbiology, and engineering domains.
This is a strong opportunity for candidates seeking roles in sterile manufacturing environments, quality systems, and technical operations within a growing pharmaceutical organization. With hiring across entry-level to managerial roles, professionals with varied experience levels can explore suitable opportunities. CEPH Lifesciences Hiring for Production QA QC
Company Overview
CEPH Lifesciences is a pharmaceutical manufacturing company focused on delivering high-quality formulations through robust manufacturing and quality systems. The company operates a modern facility in Baddi, one of India’s leading pharmaceutical hubs. With a focus on compliance, quality assurance, and operational excellence, CEPH Lifesciences continues to expand its technical teams across multiple functional areas.
The organization provides career opportunities for professionals experienced in sterile manufacturing, quality systems, and pharmaceutical engineering. CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Job Location & Employment Type
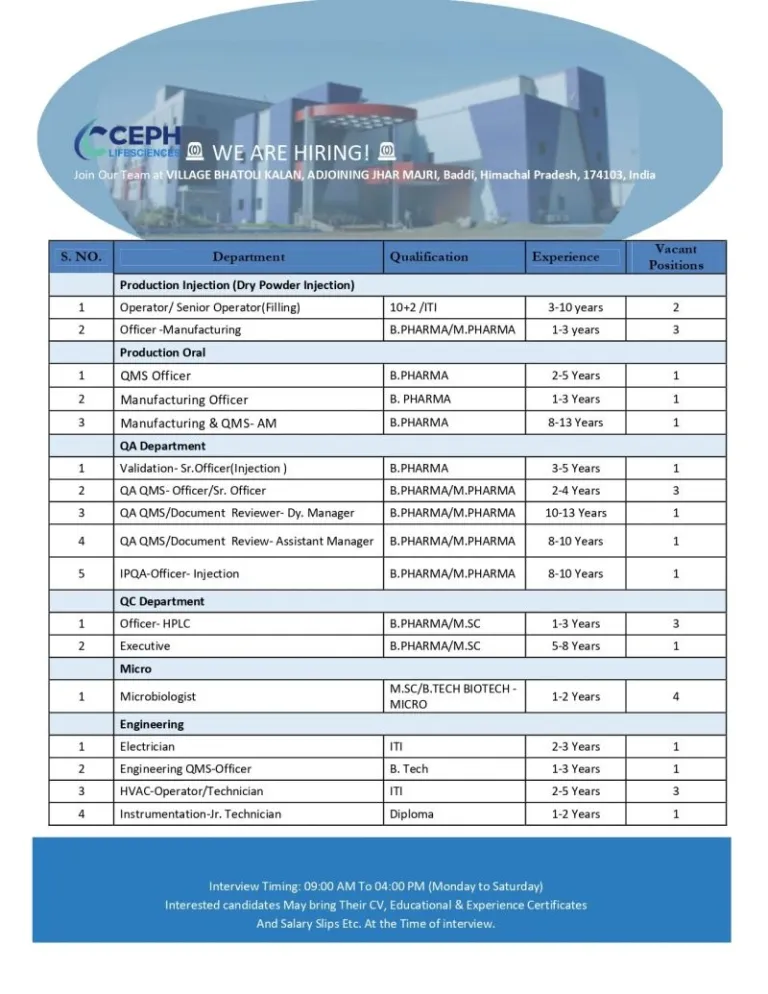
- Work Location: Baddi, Himachal Pradesh
- Employment Type: Full-time, on-site roles
- Hiring Mode: Direct walk-in / ongoing recruitment
Open Positions / Department Details
CEPH Lifesciences is recruiting across several departments, including sterile production, quality assurance, microbiology, and engineering support functions.
Production – Injection (Dry Powder Injection)
- Operators and senior operators for filling operations
- Manufacturing officers for injectable production
QMS & Manufacturing Roles
- QMS officers and manufacturing professionals
- Assistant Manager roles for experienced candidates
Quality Assurance (QA)
- Validation specialists for injectable units
- QMS professionals and documentation reviewers
- IPQA roles for sterile manufacturing environments
Quality Control (QC)
- HPLC analysts and quality testing professionals
- Senior microbiology executives
Microbiology
- Microbiologists with biotechnology or microbiology specialization
Engineering
- Electricians and HVAC technicians
- Engineering QMS officers
- Instrumentation technicians
CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Key Roles & Responsibilities
Depending on the department and seniority level, selected candidates will be responsible for: CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Production
- Managing sterile filling operations for dry powder injections
- Maintaining aseptic practices and sterile documentation
- Executing batch records and manufacturing SOPs
- Supporting process validation and troubleshooting
QMS & QA
- Managing quality management system documentation
- Reviewing validation and compliance records
- Handling document control and regulatory readiness
- Performing IPQA activities in sterile manufacturing areas
- Supporting audit preparation and quality investigations
Quality Control
- Conducting analytical testing using HPLC and related instruments
- Maintaining laboratory documentation and compliance
- Supporting method development and validation
- Ensuring data integrity and audit readiness
Microbiology
- Performing microbial testing and environmental monitoring
- Supporting sterility assurance programs
- Maintaining microbiology laboratory records
- Participating in cleanroom monitoring programs
Engineering
- Maintaining electrical and HVAC systems
- Supporting plant utilities and equipment reliability
- Managing instrumentation and calibration activities
- Ensuring compliance with engineering documentation and QMS
CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Eligibility Criteria
Education
- ITI / 10+2 for operator and technician roles
- Diploma holders for instrumentation roles
- B.Pharm / M.Pharm for production and QA positions
- M.Sc (Chemistry / Microbiology) for QC and microbiology roles
- Engineering graduates (B.Tech) for technical and engineering QMS roles
Experience
- Entry-level roles: 1–3 years
- Mid-level roles: 3–8 years
- Senior and managerial roles: Up to 13 years of experience
Skills Required
- Exposure to pharmaceutical manufacturing environments
- Knowledge of GMP and sterile manufacturing practices
- Documentation and regulatory compliance skills
- Experience in injectable facilities preferred
- Analytical and laboratory instrumentation knowledge (for QC roles)
- Strong understanding of QMS frameworks
- Technical troubleshooting skills (for engineering roles)
Salary & Benefits
Compensation will be as per company norms and aligned with industry standards based on experience and designation.
Indicative industry ranges in Himachal Pradesh may vary from entry-level packages for junior roles to significantly higher compensation for senior QA or managerial positions. Additional benefits may include performance incentives, insurance coverage, and long-term growth opportunities within the organization. CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Selection Process
The company is conducting ongoing walk-in interviews, and selection may include:
- Resume screening and document verification
- Technical evaluation by departmental experts
- HR discussion covering compensation and availability
- Final selection based on role suitability and experience
Due to continuous hiring, suitable candidates may receive quicker responses and faster onboarding timelines. CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
How to Apply
Interested candidates can directly attend walk-in interviews at the company’s Baddi facility.
Interview Venue:
CEPH Lifesciences
Village Bhatoli Kalan, Adjoining Jhar Majri
Baddi, Himachal Pradesh – 174103, India
Interview Timings:
Monday to Saturday
09:00 AM to 04:00 PM
Documents Required:
- Updated CV/resume
- Educational certificates
- Experience letters
- Salary slips (if applicable)
- Valid ID proof
Candidates should ensure all documents are complete to facilitate smooth evaluation.
Important Dates
- Interview Days: Ongoing (Monday to Saturday)
- Interview Time: 09:00 AM to 04:00 PM
Why Apply for This Pharma Job?
CEPH Lifesciences offers diverse opportunities for professionals across sterile production, quality, microbiology, and engineering domains. With hiring across junior to managerial levels, candidates can find roles aligned with their experience and specialization.
The company’s presence in Baddi provides access to one of India’s largest pharmaceutical ecosystems, offering strong industry exposure and career growth potential. Professionals working in sterile manufacturing or quality-driven environments can benefit from hands-on experience in regulated setups.
Additionally, the ongoing walk-in model provides flexibility, allowing candidates to attend interviews at convenient times rather than waiting for scheduled hiring events. CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent platform that shares pharmaceutical job updates for informational purposes only. We are not affiliated with CEPH Lifesciences or its hiring teams. Candidates should verify all job details through official sources before attending interviews. We do not charge any fees for job listings or application support. CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles
Final Call-to-Action
If you are looking for pharmaceutical job opportunities in Baddi across production, QA, QC, microbiology, or engineering functions, this ongoing hiring drive by CEPH Lifesciences is worth exploring. With multiple openings across experience levels and continuous walk-in availability, candidates can take advantage of this opportunity to secure roles in a growing pharmaceutical manufacturing organization. Prepare your documents and visit the facility during interview hours to maximize your chances of selection. CEPH Lifesciences Hiring for Production QA QC Microbiology Engineering Roles

