Remote Job ICON plc Hiring Global Study Manager for Bangalore and global locations. Apply online for remote clinical trial management role.
The global clinical research industry continues to expand as pharmaceutical companies invest heavily in developing innovative therapies and vaccines. Clinical trial management professionals play a critical role in ensuring that global studies are conducted efficiently, safely, and in compliance with regulatory standards. ICON plc, a leading global clinical research organization, is currently offering an exciting opportunity for experienced clinical research professionals.
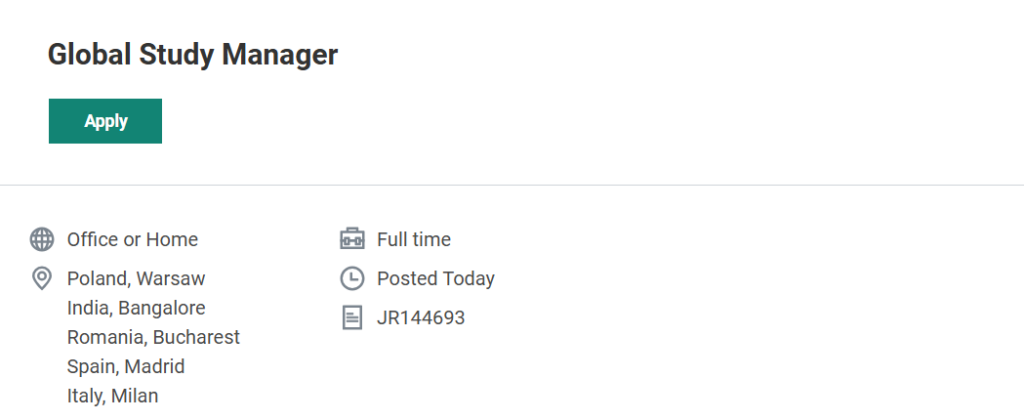
ICON is hiring a Global Study Manager (Senior Clinical Trial Manager) for a full-time position that can be performed remotely or from office locations including Bangalore. This role involves managing global clinical studies, coordinating with cross-functional teams, and ensuring that clinical trials are conducted according to regulatory and quality standards.
This position is ideal for experienced professionals who have strong clinical trial management expertise and want to contribute to global research programs focused on developing innovative medicines and vaccines. Remote Job ICON plc Hiring Global Study Manager
Company Overview
ICON plc is a globally recognized healthcare intelligence and clinical research organization that provides consulting, clinical development, and commercialization services to pharmaceutical, biotechnology, and medical device companies.
The company supports the development of new medicines and therapies by managing clinical trials across different therapeutic areas. With operations in numerous countries, ICON works with leading biopharmaceutical companies to accelerate drug development and improve healthcare outcomes worldwide.
ICON is known for its collaborative work environment, strong commitment to innovation, and focus on delivering high-quality clinical research services. Professionals working with the company gain exposure to global research programs and cutting-edge clinical development technologies. Remote Job ICON plc Hiring Global Study Manager
Job Location & Employment Type
Company: ICON plc
Position: Global Study Manager / Senior Clinical Trial Manager
Employment Type: Full-Time
Work Mode: Remote or Office-based
Available Locations:
- Bangalore, India
- Warsaw, Poland
- Bucharest, Romania
- Madrid, Spain
- Milan, Italy
Candidates can perform this role remotely or from one of the listed office locations depending on operational requirements. Remote Job ICON plc Hiring Global Study Manager
Department & Role Overview
Department: Clinical Trial Management / Global Clinical Operations
The Global Study Manager will play a key role in overseeing international clinical trials and ensuring that studies are conducted according to regulatory requirements and sponsor expectations.
The position involves coordinating with cross-functional teams, managing study timelines, monitoring trial progress, and analyzing clinical trial data to support successful research outcomes.
Professionals in this role will work closely with internal teams, sponsors, investigators, and other stakeholders involved in global clinical trials. Remote Job ICON plc Hiring Global Study Manager
Key Roles & Responsibilities
The selected candidate will manage several aspects of clinical trial execution and study coordination. Responsibilities may include:
- Collaborating with cross-functional teams to design and implement clinical trial enrollment strategies.
- Assisting in the development and implementation of clinical study protocols.
- Managing the day-to-day operational activities of global clinical trials.
- Ensuring that clinical trials comply with regulatory requirements and industry standards.
- Monitoring study progress and identifying potential challenges during the trial lifecycle.
- Analyzing clinical trial data to identify trends and provide insights that improve study outcomes.
- Implementing corrective strategies to resolve operational or study-related challenges.
- Communicating effectively with sponsors, research teams, and other project stakeholders.
- Maintaining strong relationships with internal and external partners involved in clinical trial execution.
- Ensuring project timelines, deliverables, and quality standards are achieved.
These responsibilities are essential for the successful management of complex global clinical trials. Remote Job ICON plc Hiring Global Study Manager
Eligibility Criteria
Candidates applying for the Global Study Manager role should meet the following requirements.
Educational Qualification
Applicants should have:
- A Bachelor’s degree in a relevant scientific or healthcare-related discipline
Fields such as pharmacy, life sciences, biotechnology, clinical research, or related areas are typically preferred. Remote Job ICON plc Hiring Global Study Manager
Experience Requirement
- Minimum 5 years of experience in clinical trial management
- Proven experience in managing or coordinating clinical studies
- Prior experience working in clinical research organizations or pharmaceutical companies is preferred
Skills Required
Candidates should demonstrate the following professional competencies:
- Strong understanding of clinical trial design and execution
- Knowledge of global regulatory requirements and clinical research guidelines
- Ability to analyze complex clinical data and interpret research findings
- Strong analytical and problem-solving skills
- Effective communication and stakeholder management abilities
- Excellent organizational and project management skills
- Ability to manage multiple projects simultaneously
- Attention to detail and strong documentation skills
- Experience working with global clinical research teams
Salary & Benefits
Salary: Competitive compensation package (as per company norms)
ICON offers a range of employee benefits designed to support professional growth and work-life balance. These benefits may include: Remote Job ICON plc Hiring Global Study Manager
- Competitive salary packages
- Annual leave benefits
- Comprehensive health insurance options for employees and families
- Retirement planning and savings programs
- Global Employee Assistance Program providing professional well-being support
- Life insurance coverage
- Flexible benefits depending on country policies
- Employee wellness programs
- Opportunities for professional development and career growth
Selection Process
The recruitment process for this role may include multiple stages to evaluate both technical expertise and leadership capabilities.
Typical hiring stages may include:
- Application Screening – Review of candidate qualifications and experience.
- Technical Interview – Evaluation of clinical trial management knowledge and project experience.
- Managerial Interview – Assessment of leadership skills and problem-solving abilities.
- HR Interview – Discussion regarding role expectations, work environment, and organizational culture.
Successful candidates will receive further instructions regarding onboarding and role responsibilities. Remote Job ICON plc Hiring Global Study Manager
How to Apply
Interested candidates can apply for this position through the official ICON plc careers portal.
Applicants should ensure that all information submitted during the recruitment process is accurate and complete. Providing incorrect or misleading information may result in disqualification.
Candidates can search for the position using the Job Requisition ID: JR144693 on the ICON careers website. Remote Job ICON plc Hiring Global Study Manager
Important Dates
Application Deadline: Not specifically mentioned
Candidates are encouraged to apply as early as possible because recruitment may close once suitable candidates are selected.
Why Apply for This Clinical Research Job?
Working as a Global Study Manager with ICON provides a unique opportunity to contribute to large-scale international clinical trials that help bring innovative medicines and vaccines to patients worldwide.
Key benefits of this opportunity include:
- Exposure to global clinical development programs
- Opportunity to work with leading biopharmaceutical sponsors
- Professional growth in clinical trial management
- Collaboration with international clinical research teams
- Competitive compensation and employee benefits
- Flexible work options including remote work
For experienced clinical trial professionals, this role offers strong career advancement opportunities within the global clinical research industry. Remote Job ICON plc Hiring Global Study Manager
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform that publishes pharmaceutical and clinical research job opportunities for informational purposes only. We are not affiliated with ICON plc or any other organization mentioned in this job post.
Candidates should verify all job details through official company websites before applying. Remote Job ICON plc Hiring Global Study Manager
Final Words
The Global Study Manager role at ICON plc is a valuable opportunity for experienced clinical research professionals who want to contribute to international clinical trials and advanced medical research. With flexible work options and global exposure, this position offers an excellent platform for career growth in clinical trial management.
Interested candidates should review the eligibility criteria carefully and apply through the official ICON careers portal. Remote Job ICON plc Hiring Global Study Manager

