Clinical Supplies Specialist Job at Alvotech in Bangalore with remote option. Apply now for biosimilar clinical trial supply management role.
The global biopharmaceutical industry is continuously expanding, creating new opportunities for professionals skilled in clinical research and supply management. Alvotech, a leading biosimilar-focused pharmaceutical company, is currently inviting applications for the position of Clinical Supplies Specialist within its Clinical & Medical Affairs function. This role offers professionals the opportunity to work on complex clinical trial supply operations while collaborating with international teams.
Candidates with experience in clinical trial supply chain management, biologics development, and GxP regulatory frameworks are encouraged to apply. The position offers a hybrid/partially remote working model and provides exposure to global biosimilar development programs. Clinical Supplies Specialist Job at Alvotech
Company Overview
Alvotech is an international biotechnology company dedicated to the development and manufacturing of biosimilar medicines. The organization focuses on producing high-quality, cost-effective biologic therapies that improve patient access to advanced treatments across the world.
The company operates a fully integrated model that includes research, development, manufacturing, and commercialization of biosimilars. Through its strong scientific expertise and global partnerships, Alvotech aims to accelerate the availability of affordable biologic therapies.
Within the Research and Development division, the Integrated Clinical and Medical Research (iCMR) department plays a crucial role in designing and executing clinical strategies that align with global regulatory expectations. The department manages clinical studies, supports regulatory submissions, and ensures compliance with international guidelines. Clinical Supplies Specialist Job at Alvotech
Job Location & Employment Type
- Job Role: Clinical Supplies Specialist
- Department: Clinical & Medical Affairs
- Company: Alvotech
- Work Location: Bangalore Office / India Home Office
- Work Mode: Partially Remote
- Employment Type: Full-Time
Department & Role Overview
The Clinical Supplies Specialist will play an important role in managing the planning, forecasting, and operational coordination of clinical trial supplies for biosimilar development programs. Clinical Supplies Specialist Job at Alvotech
This position involves overseeing the entire lifecycle of clinical trial material—from manufacturing planning and packaging to distribution and final disposition. The professional will collaborate with cross-functional departments such as Clinical Operations, Regulatory Affairs, and Project Management to ensure that clinical studies are adequately supplied with investigational products.
The role also requires close coordination with external vendors, contract research organizations (CROs), and logistics providers to ensure smooth supply chain operations and compliance with global regulatory standards. Clinical Supplies Specialist Job at Alvotech
Key Roles & Responsibilities
The selected candidate will be responsible for managing multiple activities related to clinical trial supply planning and operations. Major responsibilities include:
Clinical Supply Planning
- Develop comprehensive supply plans for biosimilar clinical studies based on enrollment forecasts and manufacturing timelines.
- Monitor study requirements and adjust supply forecasts according to recruitment rates and protocol changes.
- Identify potential supply risks and implement strategies to minimize disruptions during clinical trials.
Clinical Trial Material Management
- Coordinate the procurement of reference products, comparators, and ancillary items required for clinical studies.
- Oversee packaging and labeling activities in accordance with regulatory requirements across different countries.
- Manage the distribution of Investigational Medicinal Products (IMP) to clinical trial sites.
Supply Chain Operations
- Support cold chain logistics and ensure proper handling of temperature-sensitive biologic products.
- Maintain oversight of clinical supply shipments, returns, and product destruction processes.
- Track inventory levels and maintain accurate records across relevant systems.
Vendor & Stakeholder Collaboration
- Work closely with CROs, packaging vendors, and logistics partners involved in clinical supply operations.
- Participate in vendor selection processes including preparation of Request for Proposals (RFPs) and Scope of Work documentation.
- Monitor vendor performance through defined KPIs and service-level agreements.
Compliance & Documentation
- Ensure all clinical supply processes follow international regulatory guidelines including ICH-GMP and GCP.
- Maintain accurate documentation in systems such as IRT, ERP platforms, and electronic Trial Master Files (eTMF).
- Contribute to the creation and revision of Standard Operating Procedures (SOPs) and work instructions.
Strategic Collaboration
- Collaborate with clinical operations teams to align supply planning with trial timelines.
- Support budget planning and monitor operational spending for outsourced services.
- Participate in regulatory inspections and audits as a subject matter expert in clinical supply management.
Clinical Supplies Specialist Job at Alvotech
Eligibility Criteria
Candidates interested in applying for this role must meet the following educational and professional requirements.
Education
Applicants should hold a degree in one of the following disciplines:
- Pharmacy
- Biotechnology
- Life Sciences
- Biomedical Sciences
- Any other relevant scientific field
Advanced qualifications in pharmaceutical sciences or related areas may be considered an advantage. Clinical Supplies Specialist Job at Alvotech
Experience
- 2 to 5 years of professional experience in clinical trial supply management or related functions.
- Prior exposure to biologics or biosimilar drug development programs is preferred.
- Experience working with global clinical trial supply chains is beneficial.
Skills Required
Successful candidates should possess a strong combination of technical knowledge and collaboration skills, including:
- Understanding of Good Manufacturing Practices (GMP) and Good Clinical Practices (GCP).
- Familiarity with cold-chain logistics and handling of biologic drug products.
- Experience with Interactive Response Technology (IRT) systems used in clinical trials.
- Knowledge of clinical supply forecasting and inventory management tools.
- Awareness of regulatory expectations from agencies such as the FDA and EMA.
- Strong analytical, planning, and organizational skills.
- Ability to work independently while coordinating with cross-functional teams.
- Effective communication and stakeholder management abilities.
Salary & Benefits
Salary and compensation for this position will be as per company norms and will depend on the candidate’s qualifications, experience, and industry expertise. Clinical Supplies Specialist Job at Alvotech
Employees may also receive additional benefits such as:
- Exposure to global clinical research programs
- Opportunities to work on biosimilar drug development projects
- Collaborative international work environment
- Career growth within clinical research and supply chain functions
Selection Process
The recruitment process may include the following stages:
- Application Screening – Initial evaluation of candidate profiles.
- Technical Interview – Discussion on clinical trial supply management knowledge and experience.
- Managerial or HR Interview – Assessment of communication, teamwork, and role suitability.
- Final Selection – Offer issued to the selected candidate.
How to Apply
Interested candidates should submit their application through the official Alvotech careers portal before the closing date.
Applicants are advised to prepare an updated resume highlighting relevant experience in clinical trial supply management, biologics development, or pharmaceutical logistics. Clinical Supplies Specialist Job at Alvotech
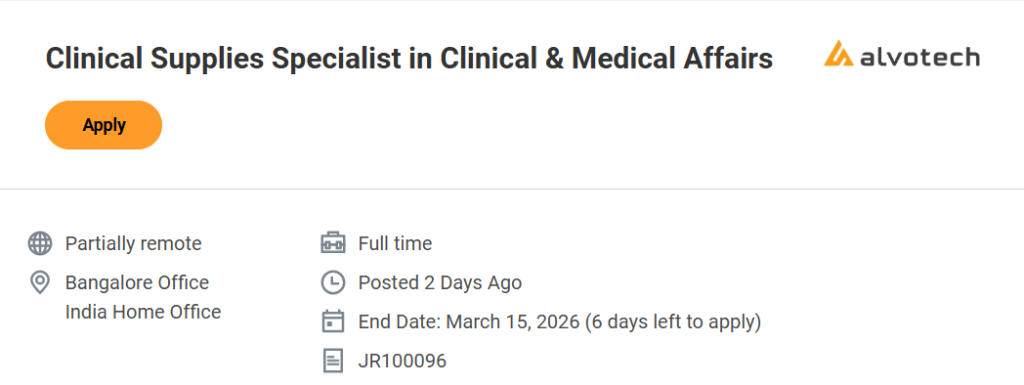
Important Dates
- Application Deadline: 15 March 2026
Candidates are encouraged to apply before the deadline as positions may be filled earlier based on suitable applications. Clinical Supplies Specialist Job at Alvotech
Why Apply for This Pharma Job?
This opportunity provides professionals with the chance to work in the rapidly growing biosimilar sector, which is transforming the pharmaceutical industry globally.
Key advantages of this role include: Clinical Supplies Specialist Job at Alvotech
- Working with a global biopharmaceutical organization
- Exposure to large-scale clinical trial supply operations
- Involvement in biosimilar drug development programs
- Opportunities to collaborate with international research teams
- Career growth within clinical research, supply chain, and regulatory operations
Professionals looking to advance their careers in clinical supply management and biosimilar development will find this role highly rewarding. Clinical Supplies Specialist Job at Alvotech
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform created to help pharmaceutical and life science professionals discover career opportunities. We are not affiliated with Alvotech or any hiring organization mentioned in this job post.
Applicants are advised to verify all information through the company’s official website before applying. We do not charge any fees for job information or recruitment services. Clinical Supplies Specialist Job at Alvotech
Final Words
If you have experience in clinical trial supply management and want to contribute to the development of biosimilar medicines, this opportunity at Alvotech could be a strong career move. Interested professionals should review the eligibility criteria and submit their application before the deadline.
Stay connected with IndiaPharmaJobs.in for the latest pharmaceutical, clinical research, and biotechnology job updates across India. Clinical Supplies Specialist Job at Alvotech

