Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles in Silvassa on 15 March 2026.
Introduction
Ipca Laboratories Limited has announced a walk-in recruitment drive for multiple manufacturing and technical roles at its pharmaceutical facility in Athal, Silvassa. The company is inviting applications from freshers and experienced professionals who want to build their careers in pharmaceutical manufacturing operations.
The recruitment drive includes opportunities in departments such as Production, Packing, Quality Assurance (QA), Quality Control (QC), Maintenance, and Utility operations. Candidates with qualifications ranging from 10th and 12th pass to ITI, Diploma, Engineering, and Graduation are eligible to participate in the interview process.
This hiring initiative offers a valuable opportunity for candidates interested in gaining experience in regulated pharmaceutical manufacturing environments and large-scale formulation production facilities. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Job Overview
| Job Title | Production, QA, QC, Maintenance, Packing Roles |
|---|---|
| Company | Ipca Laboratories Limited |
| Industry | Pharmaceutical Manufacturing |
| Job Location | Athal, Silvassa (Dadra & Nagar Haveli) |
| Employment Type | Full-Time |
| Experience Required | Freshers – 12 Years |
| Qualification | 10th, 12th, ITI, Diploma, B.Tech, B.E, Graduate |
| Interview Type | Walk-In Interview |
| Interview Date | 15 March 2026 |
| Interview Time | 9:00 AM – 4:00 PM |
| Salary | ₹1.8 LPA – ₹6 LPA (Estimated) |
Company Overview
Ipca Laboratories Limited is a well-established pharmaceutical company with a strong presence in both domestic and international pharmaceutical markets. The company manufactures a wide range of pharmaceutical formulations and active pharmaceutical ingredients (APIs) across multiple therapeutic categories.
Ipca operates advanced manufacturing facilities that comply with international regulatory standards and approvals from authorities such as USFDA, UK-MHRA, EDQM Europe, and WHO. These facilities support the company’s global supply of high-quality pharmaceutical products.
The organization focuses on maintaining strong quality systems, regulatory compliance, and modern manufacturing technologies. Employees working at Ipca Laboratories gain valuable exposure to regulated pharmaceutical operations and large-scale manufacturing environments. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Job Location & Employment Type
Job Location:
Athal, Silvassa
Dadra & Nagar Haveli, India
Employment Type:
Full-Time
Selected candidates will work at the company’s manufacturing facility located in Athal, Silvassa, which specializes in pharmaceutical formulation production. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Open Departments
Ipca Laboratories is currently recruiting professionals for the following departments:
- Production
- Packing
- Quality Assurance (QA)
- Quality Control (QC)
- Utility & Maintenance
These departments are critical to maintaining manufacturing efficiency and ensuring product quality in pharmaceutical operations.
Available Designations
Candidates may be hired for the following roles depending on their qualifications and experience:
- Operator
- Technician
- Supervisor
- Junior Officer
- Officer
- Fresher Trainee
The final designation will depend on the candidate’s technical skills, qualifications, and work experience. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Key Roles & Responsibilities
Production Department
Production professionals will be responsible for managing pharmaceutical manufacturing processes and ensuring efficient production operations. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Key responsibilities include:
- Operating pharmaceutical manufacturing equipment
- Handling processes such as granulation, compression, coating, and printing
- Maintaining production documentation and batch records
- Ensuring manufacturing activities comply with GMP standards
- Monitoring production efficiency and product quality
Packing Department
The packing department ensures that pharmaceutical products are packaged according to quality and regulatory standards.
Responsibilities include:
- Operating packaging equipment such as CAM, IMA, Blister, and VCI machines
- Maintaining packing documentation and production records
- Ensuring proper labeling and packaging of finished products
- Monitoring packaging processes for accuracy and quality compliance
Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Maintenance Department
Maintenance professionals are responsible for ensuring that manufacturing equipment operates efficiently and safely.
Key responsibilities include:
- Performing preventive maintenance of plant equipment
- Troubleshooting mechanical, electrical, and pneumatic systems
- Supporting equipment installation and calibration
- Assisting with validation and maintenance activities
Quality Assurance and Quality Control
QA and QC teams play an essential role in maintaining product quality and regulatory compliance.
Responsibilities include:
- Conducting quality control testing and analysis
- Maintaining quality documentation and reports
- Ensuring compliance with Good Manufacturing Practices (GMP)
- Supporting production teams with quality assurance processes
- Participating in quality audits and inspections
Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Utility & Maintenance Operations
Utility teams support the infrastructure required for pharmaceutical manufacturing.
Responsibilities include:
- Managing plant utilities and support systems
- Handling operations related to electrical, mechanical, instrumentation, civil, and RFM utilities
- Ensuring continuous functioning of facility infrastructure
- Maintaining equipment and system documentation
Eligibility Criteria
Educational Qualification
Candidates must possess one of the following qualifications depending on the role:
- 10th Pass
- 12th Pass
- ITI
- Diploma in Engineering
- B.Tech / B.E
- Any Graduate Degree
Relevant technical disciplines may include:
- Electrical Engineering
- Mechanical Engineering
- Instrumentation
- Civil Engineering
- RFM (Refrigeration & Maintenance)
Experience Required
Applicants may have:
- Freshers
- 0 – 12 years of experience in pharmaceutical manufacturing
Candidates with experience in tablet and capsule manufacturing operations will be given preference.
Skills Required
Candidates applying for these roles should possess:
- Basic knowledge of pharmaceutical manufacturing processes
- Understanding of GMP and quality compliance requirements
- Technical knowledge related to equipment or manufacturing operations
- Ability to work in a regulated production environment
- Good documentation and communication skills
- Willingness to work in rotational shift duties
Salary & Benefits
The salary package offered by Ipca Laboratories will depend on the candidate’s experience, qualifications, and job role.
Estimated Salary Range:
₹1.8 LPA – ₹6 LPA
Additional benefits may include:
- Performance-based incentives
- Opportunities for career advancement
- Exposure to regulated pharmaceutical manufacturing environments
- Professional development and technical training
Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Selection Process
The hiring process will be conducted through walk-in interviews.
The selection process may include:
- Document verification
- HR interaction
- Technical discussion with department representatives
- Final selection based on qualifications and experience
Candidates who successfully clear the interview process may receive job offers based on company requirements.
Walk-In Interview Details
Interview Date:
15 March 2026 (Sunday)
Interview Time:
9:00 AM – 4:00 PM
Interview Venue:
Hotel Red Maple
Jhoomer Ghat, Rasalpura
Rau, Indore
Madhya Pradesh – 453331
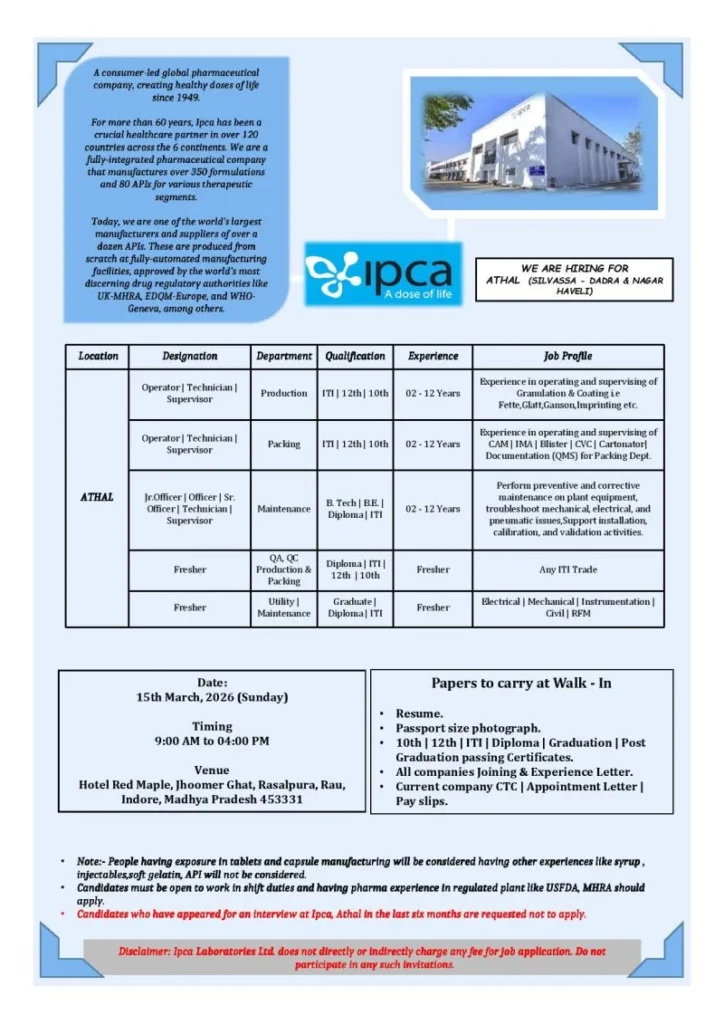
Documents Required for Interview
Candidates attending the walk-in interview should carry the following documents:
- Updated resume
- Passport-size photographs
- Educational certificates (10th / 12th / ITI / Diploma / Graduation)
- Previous company experience letters (if applicable)
- Joining letter from current or previous employer
- Latest salary slips
- Current CTC details
Important Notes
Applicants should be aware of the following conditions before attending the interview:
- Preference will be given to candidates with tablet and capsule manufacturing experience
- Candidates with experience in syrup, injectables, API, or soft gelatin manufacturing may not be considered
- Applicants must be willing to work in shift-based duties
- Candidates who attended an interview at Ipca Athal within the last six months should not apply again
Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Why Apply for This Pharma Job?
Working with Ipca Laboratories provides professionals with opportunities to build strong careers in pharmaceutical manufacturing. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Key advantages include:
- Opportunity to work in internationally regulated pharmaceutical facilities
- Exposure to tablet and capsule manufacturing processes
- Hands-on experience in production, quality control, and maintenance operations
- Career growth within a globally recognized pharmaceutical organization
- Professional development in regulated pharmaceutical environments
This recruitment drive is particularly beneficial for fresh graduates, technicians, and experienced pharmaceutical professionals seeking long-term career opportunities. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform that publishes pharmaceutical industry job updates for informational purposes only. We are not affiliated with Ipca Laboratories or any recruitment agencies.
Candidates should attend interviews or apply only through official company channels. Applicants are advised not to pay any recruitment or placement fees to individuals or agencies. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles
Final Call-to-Action
If you are interested in working in pharmaceutical manufacturing and meet the required qualifications, this walk-in opportunity at Ipca Laboratories could help advance your career. Eligible candidates are encouraged to attend the interview with all necessary documents and explore available opportunities within one of India’s leading pharmaceutical organizations. Ipca Laboratories Hiring Freshers for Production QA QC Maintenance Packing Roles

