Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job in Baddi for B.Pharm, M.Pharm, M.Sc, B.Tech candidates.
Introduction
The pharmaceutical manufacturing sector in India continues to expand, creating numerous career opportunities for skilled professionals across production, quality, and technical departments. Companies operating in regulated manufacturing environments require qualified candidates who can maintain high standards of quality, compliance, and operational efficiency.
Ceph Lifesciences has announced multiple job openings across several departments at its pharmaceutical manufacturing facility located in Baddi, Himachal Pradesh. The company is inviting applications from candidates with qualifications such as B.Pharm, M.Pharm, M.Sc, B.Tech, ITI, and other graduate degrees to join their growing team. This recruitment drive offers excellent opportunities for professionals looking to build long-term careers in pharmaceutical manufacturing. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Company Overview
Ceph Lifesciences is a pharmaceutical manufacturing company engaged in the development and production of high-quality pharmaceutical formulations. The organization focuses on maintaining strict quality standards, regulatory compliance, and advanced manufacturing practices to serve both domestic and international healthcare markets.
The company operates modern production facilities designed to meet industry standards such as Good Manufacturing Practices (GMP). Through continuous investment in infrastructure and talent development, Ceph Lifesciences aims to strengthen its presence in the pharmaceutical manufacturing industry. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Job Location & Employment Type
| Job Detail | Information |
|---|---|
| Company | Ceph Lifesciences |
| Departments | Production, QA, QC, Microbiology, Engineering, Store, EHS, Excise & Dispatch |
| Job Location | Baddi, Himachal Pradesh, India |
| Employment Type | Full-Time |
| Industry | Pharmaceutical Manufacturing |
| Experience Required | 1 – 13 Years |
| Application Mode | Walk-In Interview / Email Application |
| Job Status | Verified Opening |
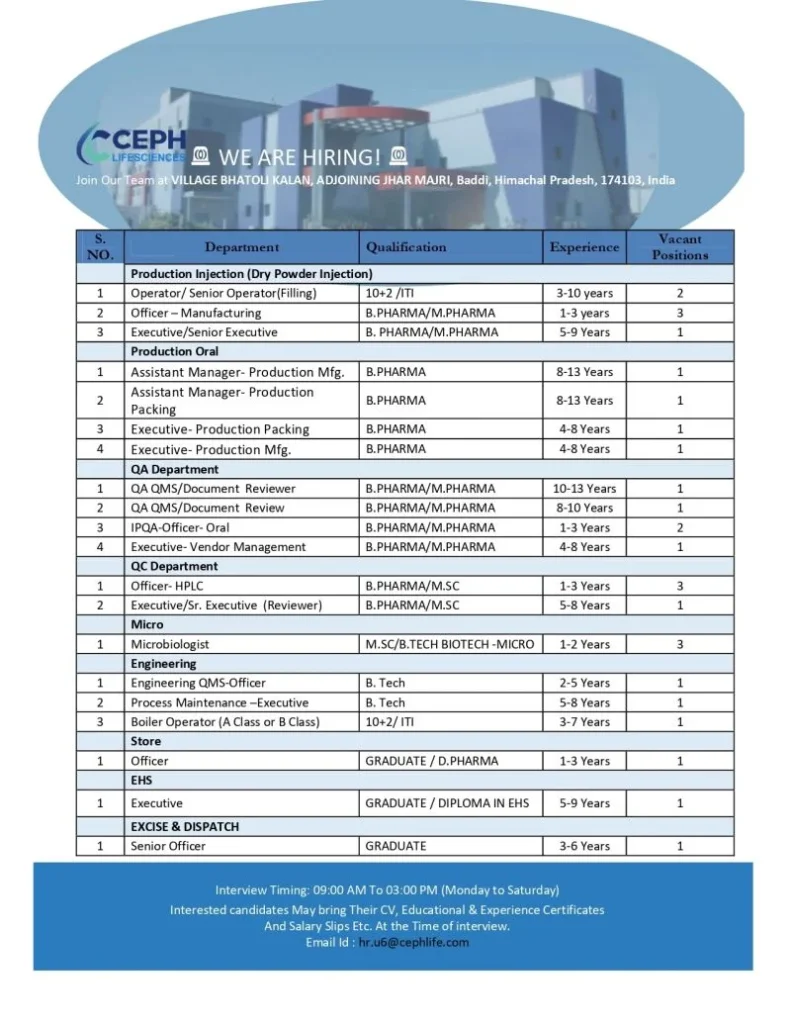
Open Positions / Department Details
Ceph Lifesciences is recruiting professionals for multiple departments including manufacturing, quality assurance, laboratory testing, and technical operations. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Production Department
Injection (Dry Powder Injection):
- Operator / Senior Operator – Filling
- Officer – Manufacturing
- Executive / Senior Executive
Oral Manufacturing:
- Assistant Manager – Production Manufacturing
- Assistant Manager – Production Packing
- Executive – Production Packing
- Executive – Production Manufacturing
Quality Assurance (QA)
- QA QMS / Document Reviewer
- IPQA Officer – Oral Manufacturing
- Executive – Vendor Management
Quality Control (QC)
- Officer – HPLC
- Executive / Senior Executive – QC Reviewer
Microbiology
- Microbiologist
Engineering
- Engineering QMS Officer
- Process Maintenance Executive
- Boiler Operator (A Class / B Class)
Store Department
- Store Officer
Environment, Health & Safety (EHS)
- EHS Executive
Excise & Dispatch
- Senior Officer
These positions provide opportunities for professionals across different technical backgrounds within pharmaceutical manufacturing operations. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Key Roles & Responsibilities
Responsibilities may vary depending on the department and job role. However, common responsibilities include:
Manufacturing & Production
- Execute pharmaceutical manufacturing processes according to Good Manufacturing Practices (GMP) guidelines.
- Operate and monitor production equipment used in pharmaceutical formulation processes.
- Maintain batch production records and manufacturing documentation.
- Ensure production processes meet regulatory and quality requirements.
Quality Assurance & Compliance
- Perform quality assurance activities to ensure compliance with regulatory standards.
- Review manufacturing documentation and maintain quality management systems.
- Conduct in-process quality checks and monitor compliance with company procedures.
Quality Control & Laboratory Operations
- Perform analytical testing using laboratory instruments such as HPLC and other analytical equipment.
- Review laboratory results and ensure compliance with quality standards.
- Maintain laboratory documentation and testing records.
Microbiology
- Conduct microbiological testing and environmental monitoring.
- Ensure microbial quality control of pharmaceutical products and manufacturing areas.
- Maintain laboratory records and support quality investigations.
Engineering & Maintenance
- Maintain and troubleshoot pharmaceutical manufacturing equipment.
- Ensure plant utilities and engineering systems operate efficiently.
- Support preventive maintenance activities and equipment validation processes.
Safety, EHS & Compliance
- Maintain workplace safety standards and ensure compliance with EHS regulations.
- Monitor environmental safety practices within the manufacturing facility.
- Support safety audits and compliance documentation.
Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Eligibility Criteria
Education
Candidates must possess relevant qualifications depending on the department:
- B.Pharm / M.Pharm
- M.Sc or B.Tech in Biotechnology, Microbiology, or related fields
- B.Tech in Engineering disciplines
- 10+2 with ITI certification for technical operator roles
- Graduate or D.Pharm for store-related roles
- Diploma in Environment Health & Safety for EHS positions
Experience
The company is hiring professionals across multiple experience levels:
- 1 – 3 years experience
- 3 – 6 years experience
- 4 – 8 years experience
- 5 – 9 years experience
- 8 – 13 years experience
Candidates with relevant pharmaceutical manufacturing experience will be preferred. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Skills Required
- Knowledge of GMP and pharmaceutical manufacturing practices
- Strong documentation and compliance awareness
- Analytical and problem-solving abilities
- Ability to work in regulated manufacturing environments
- Team collaboration and communication skills
- Experience with pharmaceutical equipment and laboratory instruments (role dependent)
Salary & Benefits
The expected salary for these roles may range between:
₹2.4 LPA – ₹10 LPA depending on the candidate’s experience, qualifications, and job role.
Additional benefits may include:
- Competitive pharmaceutical industry compensation
- Career advancement opportunities
- Exposure to regulated pharmaceutical manufacturing environments
- Professional development and learning opportunities
- Stable employment within a growing pharmaceutical organization
Selection Process
The recruitment process may include the following stages:
- Walk-In Interview / Resume Screening
- Technical Interview (Department Specific)
- HR Discussion
- Final Selection
Candidates attending the walk-in interview may be evaluated directly by the hiring team. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Walk-In Interview Details
| Interview Detail | Information |
|---|---|
| Interview Time | 09:00 AM – 03:00 PM |
| Interview Days | Monday to Saturday |
| Interview Location | Village Bhatoli Kalan, Adjoining Jhar Majri |
| City | Baddi |
| State | Himachal Pradesh |
| PIN Code | 174103 |
Documents Required
Candidates attending the interview should bring:
- Updated Resume / CV
- Educational Certificates
- Experience Certificates
- Recent Salary Slips (if applicable)
- Government-issued ID proof
How to Apply
Interested candidates can apply through either of the following methods:
Walk-In Interview
Candidates can directly attend the walk-in interview at the location mentioned above during the scheduled time.
Email Application
Applicants may also send their updated resume to:
Email: hr.u6@cephlife.com
Shortlisted candidates will be contacted by the HR team for further discussion. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Why Apply for This Pharma Job?
Baddi, Himachal Pradesh, is one of the largest pharmaceutical manufacturing hubs in India, offering significant career opportunities for pharmaceutical professionals. Working in such a regulated environment allows candidates to gain valuable experience in GMP manufacturing, quality systems, and pharmaceutical production technologies.
This recruitment drive by Ceph Lifesciences provides professionals with opportunities to work in multiple departments including production, quality control, microbiology, engineering, and safety management. Candidates seeking stable career growth within the pharmaceutical manufacturing sector can benefit from this opportunity. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent platform that shares pharmaceutical job updates for informational purposes only. We are not associated with Ceph Lifesciences or any company mentioned in this post. Candidates should verify job details from the official company sources before applying. IndiaPharmaJobs.in does not charge any fee for providing job information. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job
Final Call-to-Action
Pharmaceutical professionals looking for opportunities in manufacturing, quality assurance, quality control, microbiology, or engineering should consider attending this recruitment drive. If you meet the eligibility criteria and want to build a career in the pharmaceutical manufacturing industry, apply via email or attend the walk-in interview at Ceph Lifesciences Baddi. Ceph Lifesciences Hiring for Production QA QC Microbiology Engineering Job

