Alvotech hiring Pharmacovigilance Associate Safety Data Management in Bengaluru with remote option. Life sciences candidates with PV experience apply.
Company Overview
Alvotech is a global biopharmaceutical company dedicated to the development and manufacturing of high-quality biosimilar medicines. The company focuses exclusively on biosimilar drug development, helping expand access to affordable biological therapies for patients worldwide.
Alvotech operates advanced research and development facilities and collaborates with global healthcare partners to bring biosimilar medicines to market. Its research strategy combines scientific expertise, regulatory compliance, and modern technology to deliver high-quality biologic treatments.
Within Alvotech’s Research and Development (R&D) division, the Integrated Clinical and Medical Research (iCMR) department manages clinical studies and post-marketing activities for biosimilar products. The department is currently working on a major transformation initiative aimed at improving operational efficiency and strengthening global safety data management processes.
As part of this initiative, Alvotech is recruiting a Pharmacovigilance Associate – Safety Data Management to join its growing global safety team. Alvotech Hiring Pharmacovigilance Associate Job
Job Overview Alvotech Hiring Pharmacovigilance Associate Job
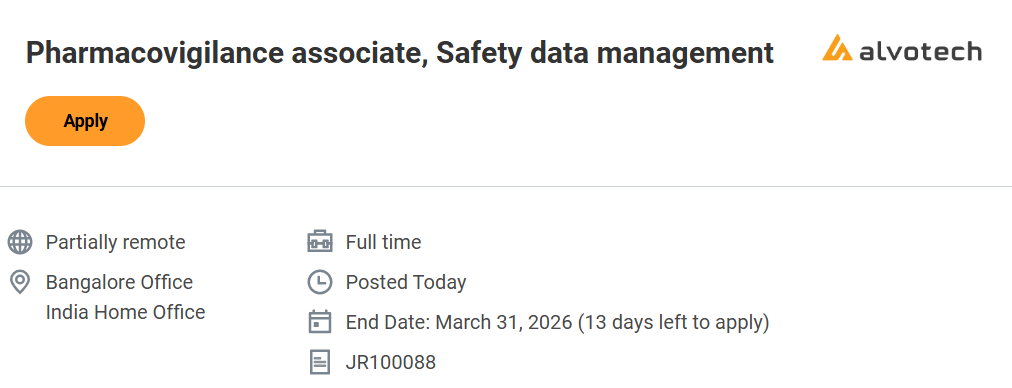
| Job Title | Pharmacovigilance Associate – Safety Data Management |
|---|---|
| Company | Alvotech |
| Job Location | Bengaluru Office / India Home Office |
| Work Model | Partially Remote |
| Employment Type | Full-Time |
| Experience Required | Minimum 2 Years |
| Qualification | Master’s Degree in Pharmacy / Life Sciences / Medicine |
| Industry | Biopharmaceutical / Pharmacovigilance |
| Application Mode | Online Application |
| Job Requisition ID | JR100088 |
Job Location & Employment Type
The selected candidate will work from Alvotech’s Bengaluru office or India home office under a partially remote working model.
This is a full-time position within the company’s Global Safety Data Management team, where the employee will contribute to pharmacovigilance operations related to clinical trials and post-marketing safety monitoring. Alvotech Hiring Pharmacovigilance Associate Job
Open Position / Department Details
Pharmacovigilance Associate – Safety Data Management
The Pharmacovigilance Associate will be responsible for supporting clinical safety and post-marketing pharmacovigilance activities across global projects. The role focuses on safety data management, regulatory compliance, and safety system operations. Alvotech Hiring Pharmacovigilance Associate Job
The selected candidate will work as part of Alvotech’s global safety data management team and report to the Safety Data Management Director.
Professionals in this role will collaborate with cross-functional teams to ensure safety data is accurately processed and maintained according to global pharmacovigilance standards.
Key Roles & Responsibilities
The Pharmacovigilance Associate will perform multiple safety data management activities within clinical and post-marketing programs. Alvotech Hiring Pharmacovigilance Associate Job
Key responsibilities include:
- Managing Individual Case Safety Reports (ICSRs) and Serious Adverse Events (SAEs) related to clinical trials and marketed products
- Supporting pharmacovigilance activities including regulatory submissions, literature monitoring, and digital platform monitoring
- Conducting safety data reconciliations and supporting compliance activities
- Managing pharmacovigilance system data such as EudraVigilance (EV) and XEVMPD submissions
- Supporting configuration, validation, and lifecycle management of safety systems
- Maintaining and improving global pharmacovigilance processes and procedures
- Ensuring compliance with international pharmacovigilance regulations including FDA, EMA, and ICH guidelines
- Collaborating with internal teams including clinical research, regulatory affairs, quality, and IT
- Coordinating with external partners such as pharmacovigilance service providers and business partners
- Delivering training sessions on pharmacovigilance procedures and compliance standards
- Supporting pharmacovigilance audits and regulatory inspections
Eligibility Criteria
Candidates interested in applying for this position should meet the following requirements. Alvotech Hiring Pharmacovigilance Associate Job
Education
Applicants should possess a Master’s degree in one of the following fields:
- Pharmaceutical Sciences
- Pharmacy
- Medicine
- Life Sciences
Experience
- Minimum 2 years of experience in Pharmacovigilance or Drug Safety
- Experience working in clinical trial safety management and post-marketing pharmacovigilance activities
Skills Required
Candidates applying for this role should ideally have:
- Practical knowledge of pharmacovigilance processes and safety data management
- Experience with safety databases such as Argus Safety, ARISg, or similar systems
- Familiarity with Veeva Safety Vault (preferred)
- Understanding of international pharmacovigilance regulations such as ICH, GVP, GCP, and CIOMS guidelines
- Experience supporting global pharmacovigilance programs and clinical trials
- Strong communication and teamwork skills
- Ability to manage multiple tasks and work independently
Alvotech Hiring Pharmacovigilance Associate Job
Preferred Experience
The following experience will be considered an advantage:
- Previous work with biologic or combination pharmaceutical products
- Experience working with global clinical trials and pharmacovigilance programs
- Exposure to safety system implementation and validation processes
Salary & Benefits
Salary and compensation for this role will be as per company norms, depending on the candidate’s qualifications and experience. Alvotech Hiring Pharmacovigilance Associate Job
Additional Benefits
Employees working with Alvotech may receive:
- Opportunity to work in a global biopharmaceutical organization
- Exposure to international pharmacovigilance operations and regulatory frameworks
- Professional development and training programs
- Collaboration with global teams and partners
- Flexible working model with partial remote work
Selection Process
The recruitment process may include the following stages:
- Online application submission
- Resume screening and candidate shortlisting
- HR interview with shortlisted candidates
- Technical interview related to pharmacovigilance and safety systems
- Final evaluation and candidate selection
Candidates who successfully complete the interview process may receive further onboarding instructions. Alvotech Hiring Pharmacovigilance Associate Job
How to Apply
Interested candidates can apply for the Pharmacovigilance Associate – Safety Data Management role through the Alvotech careers portal by submitting their updated resume.
Applicants should highlight their experience in pharmacovigilance, safety data management, and drug safety systems in their application. Alvotech Hiring Pharmacovigilance Associate Job
Important Dates
| Event | Date |
|---|---|
| Application Deadline | 31 March 2026 |
Candidates are encouraged to apply before the deadline.
Why Apply for This Pharma Job?
This role offers an excellent opportunity to work in a global biosimilar development company focused on innovative biologic medicines. Professionals joining Alvotech will gain exposure to global pharmacovigilance systems, clinical safety operations, and regulatory compliance processes.
Working in the company’s safety data management team also allows professionals to develop expertise in drug safety databases, regulatory submissions, and international pharmacovigilance standards, which are essential skills in the pharmaceutical industry. Alvotech Hiring Pharmacovigilance Associate Job
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform that shares pharmaceutical and healthcare career opportunities for informational purposes only. We are not affiliated with Alvotech or any organization mentioned in this post.
Applicants are advised to verify job details directly with the company before submitting applications or sharing personal information. Alvotech Hiring Pharmacovigilance Associate Job
Final Call-to-Action
Professionals with experience in pharmacovigilance and safety data management who want to work in a global biopharmaceutical organization should consider applying for this opportunity. If you meet the eligibility criteria, submit your application for the Pharmacovigilance Associate role at Alvotech and advance your career in drug safety. Alvotech Hiring Pharmacovigilance Associate Job

