Project Manager Remote Jobs in Clinical Research at Syneos Health. Remote and hybrid roles available. Apply for future CRO opportunities now.
Introduction
For experienced professionals in clinical research, project management roles offer strong career growth and global exposure. A leading CRO is building a talent pipeline for Project Managers across India. These roles are ideal for candidates with clinical operations experience who want to lead studies, manage teams, and work on international clinical trials.
Company Overview
Syneos Health is a globally recognized biopharmaceutical services organization that supports clinical development, medical affairs, and commercialization. The company partners with pharmaceutical and biotech firms to accelerate drug development and improve patient outcomes. With a strong global workforce and presence across 100+ countries, it offers professionals the opportunity to work on large-scale clinical research projects.
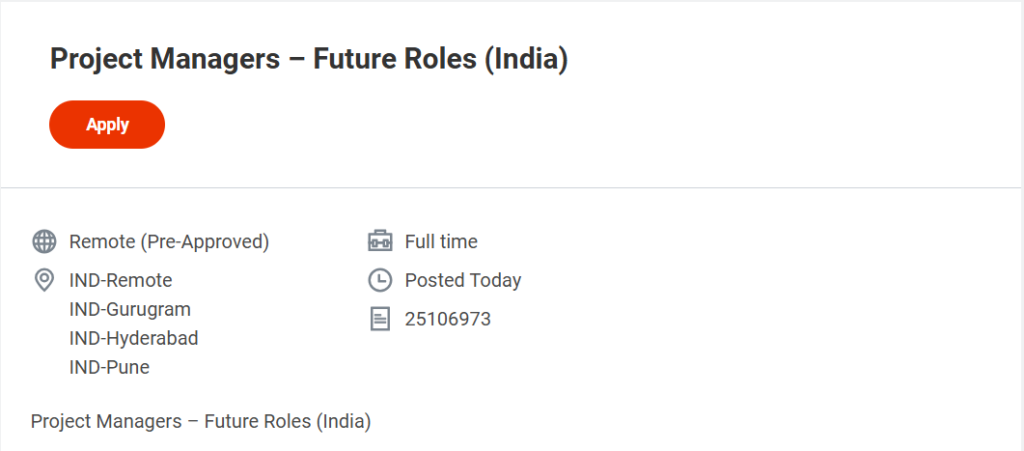
Job Overview
| Job Details | Information |
|---|---|
| Job Role | Project Manager – Clinical Research |
| Company | Syneos Health |
| Location | Remote / Hyderabad / Gurugram / Pune |
| Experience | Experienced Professionals |
| Qualification | Life Sciences / Pharmacy / Nursing |
| Employment Type | Full-Time |
| Work Mode | Remote / Hybrid |
| Job ID | 25106973 |
Job Location & Employment Type
- Locations: Remote (India), Hyderabad, Gurugram, Pune
- Work Mode: Remote (pre-approved) / Hybrid
- Employment Type: Full-time
- Travel Requirement: Up to 25%
Open Positions / Department Details
- Department: Clinical Project Management
- Role Level: Mid to Senior Level
- Hiring Type: Talent pipeline (future roles)
- Team Handling: Yes (depending on project requirements)
Key Roles & Responsibilities
As a Project Manager, you will lead clinical trials and ensure successful project delivery. Your responsibilities will include:
- Managing end-to-end clinical research projects across multiple functions
- Acting as the primary point of contact between clients and internal teams
- Ensuring studies are delivered within timelines, budget, and quality standards
- Monitoring compliance with ICH-GCP, SOPs, and regulatory requirements
- Coordinating activities across clinical operations, data management, and other teams
- Managing project finances, forecasting, and budget tracking
- Maintaining Trial Master File (TMF) quality and completeness
- Preparing project reports and presenting updates to stakeholders
- Identifying risks and implementing mitigation strategies
- Supporting inspection readiness and audit processes
- Building strong relationships with clients and contributing to business development
Eligibility Criteria
Education
- Bachelor’s degree in Life Sciences, Pharmacy, Medicine, Nursing, or related field
- Equivalent experience may also be considered
Experience
- Prior experience in clinical research or CRO industry
- Experience in project management or study management roles
- Therapeutic area expertise is an added advantage
Skills Required
Candidates should demonstrate:
- Strong knowledge of ICH-GCP and regulatory guidelines
- Project management and leadership skills
- Excellent organizational and time management abilities
- Strong communication and stakeholder management skills
- Ability to manage multiple projects simultaneously
- Financial understanding (budgeting and forecasting)
- Risk management and problem-solving capabilities
- Adaptability to new tools and technologies
Salary & Benefits
- Salary: As per company norms
- Opportunity to work on global clinical trials
- Flexible work options (remote/hybrid)
- Career development and leadership growth programs
- Exposure to cross-functional teams and international clients
- Performance-based rewards and recognition
Selection Process
The hiring process may include:
- Resume shortlisting
- Technical and project management interviews
- Client/stakeholder discussions (if required)
- HR round and final selection
How to Apply
Interested candidates can apply through the official careers portal of the company.
- Job ID: 25106973
- Apply online to be considered for upcoming opportunities
Important Dates
- Last Date: Not specified
- Note: This is a pipeline role for future hiring
Why Apply for This Pharma Job?
Project management roles are among the most rewarding career paths in clinical research. By applying for this opportunity, you can:
- Lead global clinical trials and research programs
- Work with international clients and cross-functional teams
- Gain expertise in project planning, budgeting, and execution
- Advance into senior leadership roles in the CRO industry
- Build a strong professional network in clinical research
This role is ideal for professionals aiming to transition into strategic and leadership positions.
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform. We are not affiliated with Syneos Health or its recruitment team. We only provide job updates for informational purposes. Candidates should verify details independently before applying.
Final Call-to-Action
If you have experience in clinical research and want to move into project management roles, this is a valuable opportunity. Apply now to be considered for upcoming openings and take your career to the next level.

