Research Associate R&D Job in Maharashtra at ZIM Laboratories for pharma R&D professionals with HPLC experience apply now for career growth opportunity
Advance Your Career in Analytical R&D and Method Development
If you have hands-on experience in analytical techniques like HPLC and dissolution testing, this opportunity can help you grow in pharmaceutical R&D. A reputed pharma company is hiring Research Associates for its Nagpur facility, offering exposure to global regulatory standards and method validation projects.
Company Overview
ZIM Laboratories Limited is a growing pharmaceutical organization specializing in drug delivery systems and innovative formulations. With a strong presence in regulated and semi-regulated markets, the company focuses on research-driven development and compliance with global standards such as USFDA and EU regulations.
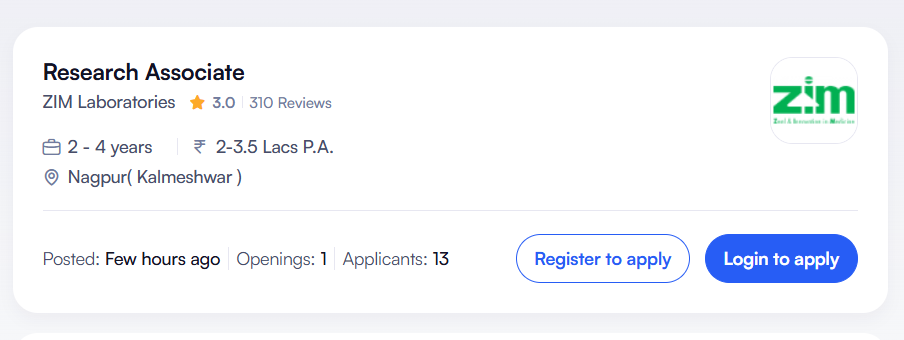
Job Overview (Quick Details)
| Field | Details |
|---|---|
| Job Title | Research Associate (R&D) |
| Company | ZIM Laboratories Limited |
| Location | Nagpur (Kalmeshwar), Maharashtra |
| Department | Analytical R&D |
| Employment Type | Full-Time, Permanent |
| Experience Required | 2–4 Years |
| Qualification | B.Pharm / M.Pharm / M.Sc (Analytical Chemistry or relevant field) |
| Salary | ₹2.5 – ₹3.5 LPA (Estimated) |
| Open Positions | 1 |
Open Positions / Department Details
This role is within the Analytical Research & Development (R&D) team. The selected candidate will work on method development, validation, and analytical testing of pharmaceutical formulations, ensuring compliance with international regulatory standards.
Key Roles & Responsibilities
As a Research Associate (Analytical R&D), your responsibilities will include:
- Perform analytical method development and validation using HPLC
- Conduct dissolution testing for pharmaceutical formulations
- Execute validation protocols in accordance with ICH guidelines
- Ensure compliance with regulatory standards such as USFDA and EU
- Maintain accurate and audit-ready documentation
- Analyze data and prepare reports for R&D projects
- Support formulation and development teams with analytical insights
Eligibility Criteria
Education
- B.Pharm / M.Pharm
- M.Sc in Analytical Chemistry or relevant discipline
Experience
- 2 to 4 years of experience in pharmaceutical R&D or analytical laboratory
- Experience in method development and validation is essential
Skills Required
Technical Skills:
- Strong expertise in HPLC (High-Performance Liquid Chromatography)
- Hands-on experience in dissolution testing
- Knowledge of analytical method validation protocols
- Familiarity with ICH Q2 guidelines
Regulatory Knowledge:
- Understanding of USFDA, EU, and ROW regulatory requirements
- Knowledge of documentation practices and compliance standards
Soft Skills:
- Analytical thinking and problem-solving ability
- Attention to detail and accuracy
- Good documentation and reporting skills
Salary & Benefits
- Salary: ₹2.5 – ₹3.5 LPA (Estimated)
- Exposure to global regulatory standards (USFDA, EU markets)
- Hands-on experience in analytical method development
- Career growth opportunities in R&D, QC, and regulatory domains
Selection Process
The selection process may include:
- Resume screening
- Technical interview (Analytical R&D / HPLC / Validation)
- HR discussion
- Final selection
How to Apply
Interested candidates should apply through the official online application link available on the company’s career portal or job listing platform.
Important Dates
- Application Deadline: Not specified (apply early)
Why Apply for This Pharma Job?
This role is ideal for professionals looking to build expertise in analytical R&D and regulatory-compliant pharmaceutical development. It offers valuable experience in method validation and exposure to global regulatory environments.
Key advantages include:
- Work in a specialized analytical R&D environment
- Gain experience with HPLC and dissolution testing
- Exposure to international regulatory standards
- Strengthen career prospects in R&D, QC, and regulatory roles
- Opportunity to work on innovative drug delivery systems
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform and is not affiliated with ZIM Laboratories Limited or any employer mentioned. Candidates are advised to verify details from official sources before applying. We do not charge any fees for job postings or applications.
Final Call-to-Action
If you have experience in analytical R&D and want to grow in pharmaceutical research, apply now. Take your career forward with this promising opportunity in Nagpur.

