Stallion Laboratories Hiring for Production QA QC Engineering Jobs in Gujarat for freshers and experienced candidates apply now via email
Mass Hiring Opportunity in Pharma Manufacturing & Quality Domains
If you are looking for a stable and growth-oriented career in the pharmaceutical industry, this is a great opportunity. A reputed pharma manufacturing company is hiring across multiple departments including Production, Quality Assurance, Quality Control, Engineering, and more. Both freshers and experienced professionals can apply for various roles based on their qualifications and expertise.
Company Overview
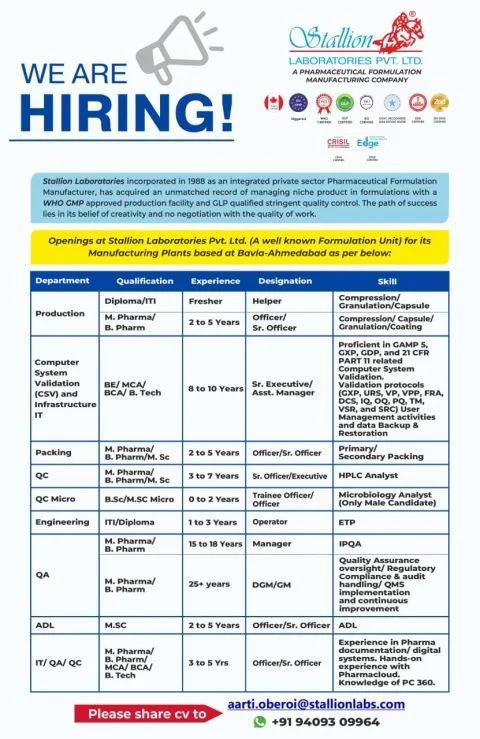
Stallion Laboratories Pvt Ltd is a well-established pharmaceutical formulation company founded in 1988. The organization operates WHO-GMP certified manufacturing facilities and is known for its strong focus on quality, compliance, and innovation. Stallion Laboratories provides professionals with exposure to regulated environments and modern pharma manufacturing practices.
Job Overview (Quick Details)
| Field | Details |
|---|---|
| Job Title | Production / QA / QC / Engineering / Packing & More |
| Company | Stallion Laboratories Pvt Ltd |
| Location | Gujarat |
| Department | Multiple (Production, QA, QC, Engineering, IT, ADL, etc.) |
| Employment Type | Full-Time |
| Experience Required | 0–10+ Years |
| Qualification | B.Pharm, M.Pharm, BSc, MSc, B.Tech, MCA, BCA, Diploma, ITI |
| Salary | ₹1.8 LPA – ₹12 LPA (Estimated) |
| Job Mode | On-site |
Open Positions / Department Details
The company is hiring across multiple departments:
- Production
- Quality Assurance (QA)
- Quality Control (QC)
- QC Microbiology
- Engineering
- Packing
- Analytical Development Lab (ADL)
- Computer System Validation (CSV) / IT Infrastructure
- IT / QA / QC Combined Roles
These roles are available for entry-level, mid-level, and senior-level candidates.
Key Roles & Responsibilities
Production
- Handle granulation, compression, and coating processes
- Ensure smooth manufacturing operations
- Follow SOPs and GMP guidelines
Quality Assurance (QA)
- Perform IPQA activities and in-process checks
- Manage audits and compliance processes
- Ensure adherence to regulatory standards
Quality Control (QC)
- Conduct HPLC and analytical testing
- Maintain product quality and consistency
- Perform laboratory documentation and reporting
Engineering
- Manage equipment maintenance and plant operations
- Ensure smooth functioning of manufacturing systems
Packing
- Handle primary and secondary packaging operations
- Maintain packaging quality standards
CSV / IT
- Manage computer system validation activities
- Ensure compliance with data integrity regulations such as 21 CFR Part 11
Eligibility Criteria
Education
Candidates must have any of the following qualifications depending on the role:
- B.Pharm / M.Pharm
- B.Sc / M.Sc
- B.Tech / MCA / BCA
- Diploma / ITI
Experience
- Freshers (for trainee and entry-level roles)
- 1–3 years (Junior roles)
- 2–5 years (Mid-level roles)
- 5–10 years (Senior roles)
- 10+ years (Manager / DGM positions)
Skills Required
Technical Skills:
- Knowledge of GMP and pharmaceutical manufacturing processes
- Understanding of QA/QC systems and documentation
- Experience with analytical techniques (HPLC preferred for QC roles)
- Familiarity with regulatory compliance and audits
Soft Skills:
- Good communication and teamwork abilities
- Attention to detail and quality focus
- Problem-solving and analytical thinking
- Ability to work in a structured manufacturing environment
Salary & Benefits
- Salary Range: ₹1.8 LPA – ₹12 LPA (depending on role and experience)
- Opportunity to work in a WHO-GMP approved facility
- Exposure to regulatory and quality systems
- Career growth in a reputed pharmaceutical company
- Professional and structured work environment
Selection Process
The hiring process may include:
- Resume screening
- Technical interview (department-specific)
- HR discussion
- Final selection
How to Apply
Interested candidates can apply via email:
📧 Send your updated CV to: aarti.oberoi@stallionlabs.com
📞 Contact: +91 94093 09964
📌 Mention your preferred department and position in the email subject line.
Important Dates
- Application Deadline: Not specified (apply early)
Why Apply for This Pharma Job?
This is a large-scale hiring opportunity offering roles across multiple departments, making it suitable for candidates at different career stages.
Key advantages include:
- Opportunities for both freshers and experienced professionals
- Exposure to GMP-compliant manufacturing environment
- Career growth across multiple pharma domains
- Hands-on experience in production, quality, and engineering
- Stable job opportunities in a reputed pharma organization
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform and is not affiliated with Stallion Laboratories Pvt Ltd or any employer mentioned. Candidates are advised to verify details from official sources before applying. We do not charge any fees for job postings or applications.
Final Call-to-Action
If you are looking for opportunities in pharma manufacturing, QA, QC, or engineering, apply now. Take advantage of this hiring drive and grow your career in the pharmaceutical industry.

