Apply for Parexel Senior Data Engineer remote job in India. Check eligibility, data visualization skills, experience requirements, and application details now.
Introduction
Professionals with expertise in clinical data analytics, visualization tools, and programming now have an exciting opportunity to work with a globally recognized CRO. This role focuses on developing data visualizations for medical review and safety signal detection, making it ideal for candidates looking to advance in clinical data engineering and analytics within the pharmaceutical industry.
Company Overview
Parexel is a leading global Contract Research Organization (CRO) that delivers clinical development, regulatory consulting, and data-driven solutions to pharmaceutical, biotechnology, and medical device companies. The company plays a crucial role in accelerating drug development and ensuring high-quality clinical trial outcomes worldwide.
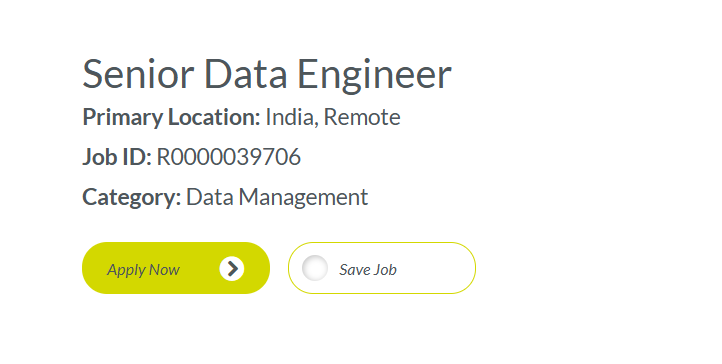
Job Overview (Quick Details)
| Job Title | Senior Data Engineer |
|---|---|
| Company | Parexel |
| Location | India (Remote) |
| Employment Type | Full-Time |
| Department | Data Management / Clinical Data Analytics |
| Experience Required | 5+ Years |
| Education | Computer Science / IT / Related Field |
| Salary | As per company norms |
| Job ID | R0000039706 |
Job Location & Employment Type
- Location: Remote (India)
- Employment Type: Full-time
Open Positions / Department Details
- Department: Data Management & Visual Analytics
- Focus on clinical data visualization and safety signal detection
- Work closely with medical and data science teams
Key Roles & Responsibilities
Data Visualization & Analytics
- Develop and maintain standardized dashboards and visualizations for medical data review
- Support safety signal detection through visual analytics
- Apply visualization standards aligned with RBQM (Risk-Based Quality Management) principles
- Ensure outputs align with Critical to Quality (CtQ) endpoints
Data Engineering & Programming
- Utilize tools like TIBCO Spotfire for visualization development
- Work with R programming and associated packages for data analysis
- Support data processing and analytics workflows
- Apply SQL and Python knowledge for data handling (preferred)
Project Execution & Timelines
- Deliver visualization outputs aligned with study milestones such as First Patient In (FPI)
- Manage assigned tasks within defined timelines
- Prioritize work based on project needs and deadlines
Quality Control & Documentation
- Ensure quality and accuracy of all data visualizations
- Maintain proper documentation for traceability and compliance
- Participate in peer reviews and quality checks
Collaboration & Stakeholder Support
- Work with cross-functional teams including medical, clinical, and analytics teams
- Address stakeholder queries related to data and visualizations
- Participate in meetings and provide updates on deliverables
Learning & Development
- Attend training sessions to enhance technical and domain knowledge
- Incorporate feedback from senior team members
- Contribute to mentoring and knowledge-sharing activities
Eligibility Criteria
Education
- Bachelor’s degree in Computer Science, Information Technology, or related field
- Equivalent technical qualification may also be considered
Experience
- Minimum 5 years of experience in:
- Data visualization
- Clinical data analytics
- Data engineering roles
- Strong experience with TIBCO Spotfire and R programming
- Experience in clinical research domain is required
Skills Required
- Expertise in TIBCO Spotfire dashboards and visualization tools
- Strong programming skills in R (R packages)
- Knowledge of Python and SQL is an added advantage
- Understanding of RBQM and clinical trial processes
- Strong analytical and problem-solving skills
- Ability to handle moderately complex data challenges
- Good communication and teamwork skills
- Ability to work independently and meet deadlines
Salary & Benefits
- Salary: As per company norms
- Additional benefits may include:
- Work-from-home flexibility
- Exposure to global clinical data projects
- Career growth in data engineering and analytics
- Continuous learning and development opportunities
Selection Process
The hiring process may include:
- Application screening
- HR discussion
- Technical interview (data visualization/programming)
- Final interview
How to Apply
- Candidates should apply via the official Parexel careers portal
- Search using Job ID: R0000039706
- Submit a resume highlighting experience in data visualization and clinical analytics
Important Dates
- Last Date to Apply: Not specified
Why Apply for This Pharma Job?
- Opportunity to work on cutting-edge clinical data analytics projects
- Exposure to advanced visualization tools and global datasets
- Work with a globally recognized CRO
- Remote work flexibility
- Strong career growth in data engineering within the pharma industry
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform and is not affiliated with Parexel or any recruiter. We do not charge any fees for job postings. Candidates should verify all details through official sources before applying.
Final Call-to-Action
Interested and eligible candidates are encouraged to apply as soon as possible to take advantage of this excellent opportunity in clinical data engineering.

