Ajanta Pharma Hiring for QA QC Production Engineering Roles in Guwahati for experienced pharma manufacturing candidates. Apply via email.
Pharmaceutical professionals seeking mid-level manufacturing opportunities can explore this latest hiring update from Ajanta Pharma. The company is recruiting experienced candidates for multiple departments including Quality Assurance, Quality Control, Production, and Engineering at its Guwahati facility. These roles are ideal for professionals with experience in regulated pharma environments, especially sterile and OSD manufacturing.
If you have hands-on experience in pharma manufacturing operations, quality systems, or plant maintenance, this opportunity offers strong career growth within a reputed specialty pharma company. Ajanta Pharma Hiring for QA QC Production Engineering Roles
Company Overview
Ajanta Pharma is a well-established Indian pharmaceutical company known for its focus on specialty therapeutics such as ophthalmology, dermatology, cardiology, and pain management. The company has a strong global footprint and exports products to multiple regulated and emerging markets.
Ajanta Pharma operates advanced manufacturing facilities compliant with global regulatory standards and is recognized for its quality-driven culture and innovation-led growth. The Guwahati plant plays a key role in sterile ophthalmic and oral dosage manufacturing. Ajanta Pharma Hiring for QA QC Production Engineering Roles
Job Location & Employment Type
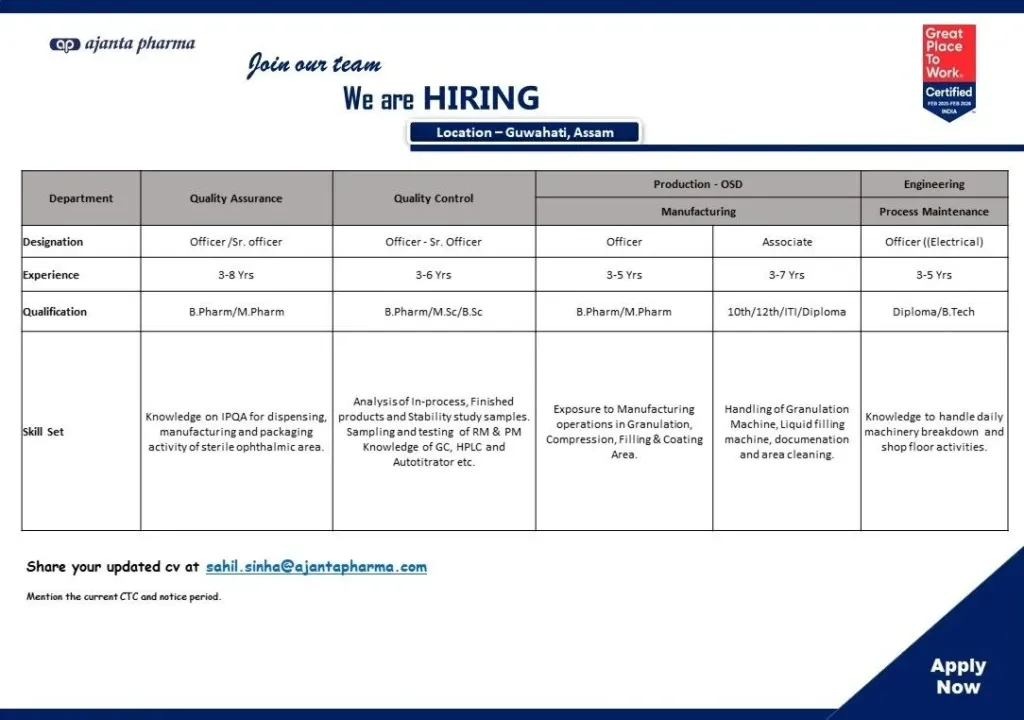
- Location: Guwahati, Assam
- Employment Type: Full-Time
- Experience Required: 3–8 Years
- Industry: Pharmaceutical Manufacturing
All positions are based at Ajanta Pharma’s manufacturing facility in Northeast India. Ajanta Pharma Hiring for QA QC Production Engineering Roles
Open Positions & Department Details
Ajanta Pharma is hiring across multiple technical departments as detailed below:
1. Quality Assurance (Sterile Ophthalmic Area)
Designation: Officer / Senior Officer
Experience: 3–8 years
Qualification: B.Pharm / M.Pharm (preferred)
Key Responsibilities:
- Handling IPQA activities in sterile manufacturing areas
- Monitoring dispensing, manufacturing, and packaging operations
- Ensuring compliance with GMP and sterile production standards
- Managing documentation and quality system adherence
Candidates with sterile ophthalmic manufacturing experience will be preferred. Ajanta Pharma Hiring for QA QC Production Engineering Roles
2. Quality Control
Designation: Officer / Senior Officer
Experience: 3–6 years
Qualification: B.Pharm / M.Pharm / M.Sc / B.Sc
Key Responsibilities:
- Analysis of raw materials, packing materials, and finished products
- In-process testing and stability study evaluations
- Sampling and documentation activities
- Handling analytical instruments such as HPLC, GC, Autotitrator, etc.
Instrumentation experience is essential for this role. Ajanta Pharma Hiring for QA QC Production Engineering Roles
3. Production – OSD Manufacturing
Designations: Officer / Associate
Experience: 3–7 years
Qualification:
- Officer: B.Pharm / M.Pharm
- Associate: Diploma / ITI / 10th or 12th
Key Responsibilities:
- Handling granulation, compression, coating, and liquid filling operations
- Managing batch manufacturing and documentation
- Supporting cleaning and compliance activities
- Ensuring adherence to manufacturing SOPs and GMP standards
Experience in oral solid dosage (OSD) manufacturing is required. Ajanta Pharma Hiring for QA QC Production Engineering Roles
4. Engineering (Electrical – Process Maintenance)
Designation: Officer
Experience: 3–5 years
Qualification: Diploma or B.Tech in Electrical Engineering (preferred)
Key Responsibilities:
- Handling equipment breakdown maintenance
- Supporting shop floor electrical operations
- Maintaining granulation and liquid filling machinery
- Preventive maintenance and equipment troubleshooting
This role is suitable for candidates with pharma plant engineering experience. Ajanta Pharma Hiring for QA QC Production Engineering Roles
Eligibility Criteria
Education
Eligible qualifications vary by role:
- B.Pharm / M.Pharm (QA, QC, Production Officer roles)
- B.Sc / M.Sc (QC roles)
- Diploma / ITI (Production Associate, Engineering)
- B.Tech (Engineering roles preferred)
Experience
- Minimum 3 years of relevant pharma industry experience
- Up to 8 years depending on role and designation
- Experience in regulated manufacturing environments preferred
Candidates with sterile, ophthalmic, or OSD exposure will have an advantage.
Skills Required
Applicants should demonstrate:
- Strong knowledge of GMP and regulatory compliance
- Hands-on pharma manufacturing or analytical experience
- Documentation and quality awareness
- Instrumentation knowledge (for QC roles)
- Troubleshooting and maintenance skills (Engineering roles)
- Ability to work in regulated and audit-ready environments
Salary & Benefits
- Estimated Salary Range: ₹3.5 – ₹8 LPA (Role and experience dependent)
- Final compensation will vary based on candidate profile and negotiation
Additional benefits may include:
- Health insurance and statutory benefits
- Performance incentives
- Growth opportunities in a specialty pharma company
- Exposure to regulated manufacturing standards
Selection Process
The recruitment process is expected to include:
- Resume shortlisting based on domain experience
- HR screening or telephonic interaction
- Technical interview with department teams
- Final discussion and offer rollout
Candidates may be evaluated on technical knowledge, practical exposure, and regulatory understanding. Ajanta Pharma Hiring for QA QC Production Engineering Roles
How to Apply
Interested candidates should apply by sending their updated CV to the official email below:
Application Tip:
Mention your current CTC and notice period in the email subject or body to improve shortlisting chances.
Important Dates
- Application Deadline: Not specified
Early applications are recommended due to limited openings.
Why Apply for This Pharma Manufacturing Job?
This opportunity is ideal for mid-level professionals seeking growth in pharmaceutical manufacturing. Key benefits include:
- Work with a reputed specialty pharma company
- Opportunities across QA, QC, Production, and Engineering
- Exposure to sterile ophthalmic and OSD manufacturing
- Career growth in a globally expanding pharma organization
- Opportunities in Northeast India with stable plant operations
Professionals aiming to build long-term careers in regulated pharma manufacturing will find this role highly valuable.
Important Note
IndiaPharmaJobs.in is an independent job information platform that shares verified pharmaceutical job updates for informational purposes only. We are not affiliated with Ajanta Pharma or any organization mentioned above. Candidates should verify all details through official company channels before applying. We do not charge any fees for job updates or recruitment information. Ajanta Pharma Hiring for QA QC Production Engineering Roles
Final Words
If you are an experienced pharma professional looking for opportunities in QA, QC, Production, or Engineering, this Ajanta Pharma hiring drive in Guwahati is worth exploring. With roles across multiple departments and strong growth potential, this opportunity can help advance your career in regulated pharmaceutical manufacturing.
Stay connected with IndiaPharmaJobs.in for the latest pharma manufacturing jobs, walk-in drives, QC openings, and industry opportunities across India. Ajanta Pharma Hiring for QA QC Production Engineering Roles

