Clinical Research Associate Job at Sun Pharma in Hyderabad for clinical trials monitoring and site management roles.
Introduction
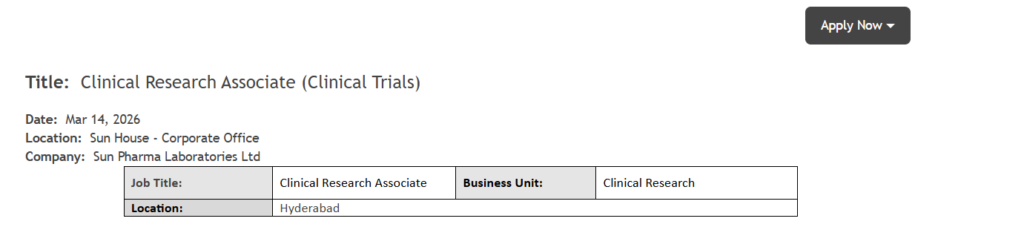
Professionals seeking opportunities in the clinical research industry can explore this exciting opening at Sun Pharma Laboratories Ltd. The company is currently hiring for the role of Clinical Research Associate (CRA) within its Clinical Research division. Clinical Research Associate Job at Sun Pharma
This position is suitable for candidates with experience in clinical trial operations, site monitoring, and regulatory compliance. The selected candidate will support clinical studies by coordinating with investigators, monitoring trial progress, ensuring adherence to regulatory guidelines, and maintaining high standards of data integrity.
Working with Sun Pharma provides an opportunity to contribute to clinical development programs that support the advancement of pharmaceutical therapies and patient care. Clinical Research Associate Job at Sun Pharma
Company Overview
Sun Pharma Laboratories Ltd is one of the world’s leading pharmaceutical companies specializing in the development and manufacturing of generic medicines and specialty pharmaceuticals. The organization operates in numerous global markets and focuses on delivering high-quality medicines that improve patient health outcomes.
The company places strong emphasis on innovation, research, and regulatory compliance. Its clinical research teams play an important role in evaluating the safety and effectiveness of pharmaceutical products through well-designed clinical trials conducted in collaboration with healthcare professionals and research institutions.
Sun Pharma promotes a culture of continuous learning, professional development, and collaboration, encouraging employees to take initiative and contribute to healthcare advancements. Clinical Research Associate Job at Sun Pharma
Job Overview
| Job Details | Information |
|---|---|
| Company Name | Sun Pharma Laboratories Ltd |
| Job Role | Clinical Research Associate (CRA) |
| Department | Clinical Research |
| Industry | Pharmaceutical / Clinical Research |
| Job Location | Hyderabad, India |
| Employment Type | Full-Time |
| Experience Required | 1 – 5 Years |
| Qualification | Bachelor’s or Master’s Degree in Health or Life Sciences |
| Application Mode | Online Application |
Job Location & Employment Type
The Clinical Research Associate position is based in Hyderabad, where the candidate will work as part of Sun Pharma’s clinical research team.
This is a full-time role involving clinical trial monitoring, investigator coordination, regulatory documentation, and data verification activities. The position may involve travel to clinical trial sites to monitor study progress and ensure compliance with study protocols. Clinical Research Associate Job at Sun Pharma
Open Positions / Department Details
Position: Clinical Research Associate
Department: Clinical Research
The Clinical Research department is responsible for planning, executing, and monitoring clinical studies that evaluate the safety and effectiveness of pharmaceutical products. Clinical Research Associates play a key role in ensuring that clinical trials are conducted according to approved protocols and regulatory requirements. Clinical Research Associate Job at Sun Pharma
Key Roles & Responsibilities
Clinical Trial Site Identification
The CRA will assist in identifying potential clinical trial sites and investigators who can participate in research studies. This includes evaluating site feasibility and coordinating with investigators during the study initiation process. Clinical Research Associate Job at Sun Pharma
Contract and Documentation Management
The candidate will support the preparation and execution of study-related agreements such as confidentiality agreements and investigator contracts. Ensuring that necessary documentation is completed before study initiation is an important responsibility.
Ethics Committee Submissions
The role includes preparing and submitting required documents to ethics committees for approval before initiating clinical studies at research sites.
Investigational Product Management
Clinical Research Associates will oversee investigational product (IP) dispensing activities and maintain accurate records related to inventory management and reconciliation.
Site Initiation and Monitoring
The candidate will coordinate site initiation activities and monitor study progress through periodic site visits. Monitoring includes verifying study procedures, ensuring protocol adherence, and documenting site activities.
Training for Investigators and Site Staff
CRAs are responsible for providing training to investigators and site personnel regarding the study protocol, clinical procedures, and Good Clinical Practice (GCP) guidelines.
Participant Recruitment Monitoring
The role includes monitoring recruitment progress at study sites and ensuring that trial participants are enrolled according to protocol requirements.
Data Verification and Query Resolution
The CRA will perform source data verification (SDV) and ensure accurate data entry in clinical trial databases. They will also assist in resolving data queries during the trial process.
Safety Reporting
Ensuring timely reporting of Serious Adverse Events (SAEs) and Suspected Unexpected Serious Adverse Reactions (SUSARs) is a critical responsibility. The CRA will coordinate with investigators and pharmacovigilance teams to maintain regulatory compliance.
Risk Management and CAPA Implementation
The role involves identifying potential risks in clinical trial operations and implementing Corrective and Preventive Actions (CAPA) when sites do not meet expected performance standards.
Cross-Functional Coordination
The Clinical Research Associate will collaborate with internal teams and external partners such as Contract Research Organizations (CROs) for activities including data management, statistical analysis, and database lock processes. Clinical Research Associate Job at Sun Pharma
Eligibility Criteria
Candidates interested in this role should meet the following eligibility requirements.
Education
Applicants must possess one of the following qualifications:
- Bachelor’s degree in Pharmacy, Biology, Chemistry, Nursing, Public Health, or other health-related disciplines
- Master’s degree in Life Sciences or a related field
- Postgraduate Diploma in Clinical Research (PGDCR) may also be considered
Experience
- Minimum 1 to 5 years of experience in clinical research or clinical trial monitoring
- Experience working with clinical trial sites or research institutions is preferred
Skills Required
Candidates should demonstrate the following competencies:
- Knowledge of Good Clinical Practice (GCP) guidelines
- Understanding of regulatory requirements for clinical trials
- Experience with Phase III and Phase IV clinical trials
- Strong site management and monitoring skills
- Ability to coordinate with investigators and cross-functional teams
- Excellent documentation and reporting abilities
- Strong communication and interpersonal skills
Clinical Research Associate Job at Sun Pharma
Salary & Benefits
The salary for this position has not been publicly disclosed and will be as per company norms.
Employees working in clinical research roles at Sun Pharma may benefit from:
- Opportunities for professional growth in clinical research
- Exposure to global clinical development programs
- Collaborative work environment with experienced professionals
- Learning opportunities in regulatory compliance and clinical trial management
Selection Process
The recruitment process may involve the following stages:
- Online Application Submission
- Profile Screening by HR Team
- Technical Interview
- HR Discussion
- Final Selection
Shortlisted candidates will be contacted by Sun Pharma’s recruitment team. Clinical Research Associate Job at Sun Pharma
How to Apply
Candidates interested in the Clinical Research Associate position at Sun Pharma can apply online through the company’s official careers portal. Clinical Research Associate Job at Sun Pharma
Applicants should ensure that their resumes highlight:
- Clinical trial monitoring experience
- Knowledge of GCP and regulatory guidelines
- Experience in site management and investigator coordination
- Skills related to data verification and clinical documentation
Providing detailed information about clinical research experience can increase the chances of being shortlisted.
Important Dates
- Last Date to Apply: Not specified by the company
Candidates are encouraged to apply as soon as possible.
Why Apply for This Pharma Job?
Clinical Research Associates play a vital role in the development of new medicines and therapies. This role provides hands-on experience in clinical trial management, regulatory compliance, and collaboration with healthcare professionals.
Working at Sun Pharma offers exposure to large-scale clinical studies and the opportunity to contribute to pharmaceutical research that impacts patient health globally. Professionals in this role can build strong careers in clinical operations, regulatory affairs, pharmacovigilance, and clinical project management. Clinical Research Associate Job at Sun Pharma
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform that publishes pharmaceutical and healthcare job updates for informational purposes only.
We do not conduct recruitment or charge any fees for job applications. Candidates should verify all job details directly with the official company website before applying. IndiaPharmaJobs.in is not affiliated with Sun Pharma Laboratories Ltd or any organization mentioned in this job posting. Clinical Research Associate Job at Sun Pharma
Final Call-to-Action
Candidates with experience in clinical research and clinical trial monitoring should consider applying for this opportunity. Submit your application through the official Sun Pharma careers portal and ensure your resume highlights expertise in site monitoring, GCP compliance, and clinical documentation to improve your chances of being shortlisted for the Clinical Research Associate role. Clinical Research Associate Job at Sun Pharma

