Clinical Studies Support Coordinator Remote Job at Medtronic. Remote clinical research role with data coordination and documentation responsibilities in India.
Are you looking to build or advance your career in clinical research and study coordination? This remote opportunity with Medtronic offers hands-on experience in clinical data management, documentation, and study support activities. If you have prior experience in clinical operations or data coordination, this role can help you grow within a global healthcare technology organization.
Company Overview
Medtronic is a leading global healthcare technology company dedicated to improving lives through innovative medical solutions. With operations in over 150 countries, the company focuses on developing advanced therapies and devices that address critical health challenges. Medtronic promotes a culture of innovation, collaboration, and continuous learning, making it a preferred employer in the healthcare industry.
Job Overview
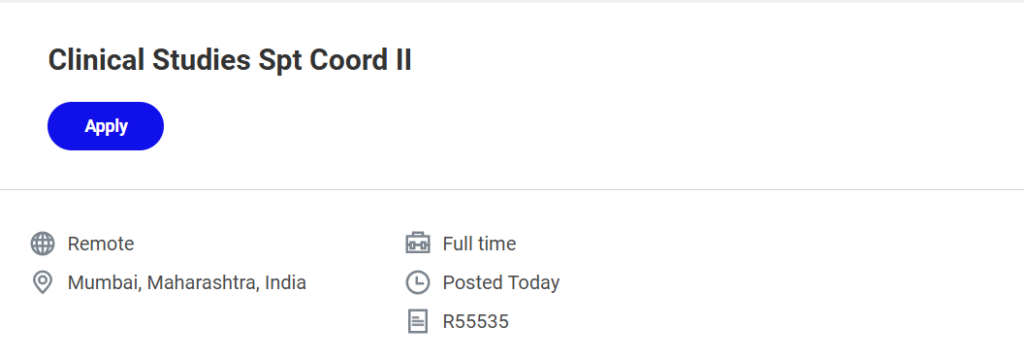
| Job Title | Clinical Studies Support Coordinator II |
|---|---|
| Company | Medtronic |
| Location | Mumbai, Maharashtra, India |
| Employment Type | Full-Time |
| Department | Clinical Research / Study Support |
| Experience | Minimum 2 Years |
| Qualification | Relevant Degree in Life Sciences / Healthcare |
| Work Mode | Remote |
| Job Requisition ID | R55535 |
Job Location & Employment Type
- Work Location: Mumbai (Remote role across India)
- Job Type: Full-time
- Work Mode: Remote / Work-from-home
Open Positions / Department Details
This role falls under the Clinical Studies Support function, focusing on providing administrative, operational, and data-related support for clinical trials. The position plays a crucial role in ensuring smooth execution and documentation of clinical studies.
Key Roles & Responsibilities
As a Clinical Studies Support Coordinator II, you will contribute to clinical trial operations by supporting data and document management processes.
Clinical Data Coordination
- Assisting in the development and testing of case report forms (CRFs) and study databases
- Supporting the preparation of study reports based on protocol requirements
- Ensuring timely and accurate completion of clinical data forms
- Verifying patient and study data for accuracy and completeness
- Identifying and resolving data discrepancies in collaboration with teams
Documentation & File Management
- Creating, organizing, and maintaining clinical study documentation
- Managing study files and ensuring proper distribution of documents
- Conducting periodic audits to ensure completeness and compliance of study records
Operational & Administrative Support
- Providing administrative support to clinical study teams
- Assisting in compensation processing and resolving discrepancies if required
- Supporting day-to-day study coordination activities
- Ensuring smooth communication across teams
Quality & Compliance
- Maintaining adherence to study protocols and regulatory standards
- Supporting internal audits and quality checks
- Identifying areas of improvement and suggesting process enhancements
Team Support & Collaboration
- Communicating with internal stakeholders across departments
- Providing guidance to junior or entry-level team members when needed
- Working collaboratively to meet project timelines and objectives
Eligibility Criteria
Educational Qualification
Candidates should have a degree in:
- Life Sciences
- Pharmacy
- Biotechnology
- Nursing
- Or any related healthcare field
Experience Required
- Minimum 2 years of relevant experience in:
- Clinical research
- Clinical data management
- Study coordination
- CRO or healthcare environment
Skills Required
- Good understanding of clinical research processes
- Strong attention to detail and data accuracy
- Basic knowledge of clinical documentation and CRFs
- Good communication and coordination skills
- Ability to manage routine tasks with minimal supervision
- Organizational and time management skills
- Problem-solving ability for handling discrepancies
- Familiarity with MS Office tools
Salary & Benefits
- Salary will be as per company norms
- Additional benefits include:
- Flexible work environment (remote role)
- Performance-based incentives (MIP)
- Health and wellness programs
- Learning and development opportunities
- Exposure to global clinical studies
Selection Process
The hiring process may include:
- Resume screening
- HR interaction
- Technical/functional interview
- Final selection round
How to Apply
Interested candidates should apply through the official Medtronic careers portal.
- Ensure your resume highlights relevant clinical or data coordination experience
- Apply early to increase your chances of selection
Important Dates
- Application deadline not explicitly mentioned
- Early application is recommended
Why Apply for This Pharma Job?
This role is ideal for candidates looking to strengthen their career in clinical research operations. Key advantages include:
- Opportunity to work in a remote clinical research role
- Hands-on experience in clinical data and documentation management
- Exposure to global clinical trial processes
- Work with a reputed healthcare technology company
- Career growth in clinical operations and data management
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform. We are not affiliated with Medtronic or any employer. We do not charge any fees for job applications. Candidates are advised to verify job details from the official website before applying.
Final Call-to-Action
If you have experience in clinical research or data coordination and are looking for a remote opportunity, apply now and grow your career with Medtronic.

