Gepach Hiring for QA QC FD Production Engineering Roles in Pithampur on 15 March 2026.
Gepach International Walk-In Interview 2026 for QA, QC, F&D, Production & Engineering Roles
Introduction
Professionals looking to build a career in pharmaceutical manufacturing have a valuable opportunity with Gepach International. The company has announced a walk-in recruitment drive for several departments including Quality Control, Quality Assurance, Formulation & Development, Production, and Engineering.
This hiring event is designed to attract both fresh graduates and experienced professionals from pharmaceutical and technical backgrounds. Candidates who meet the required qualifications are invited to attend the walk-in interview scheduled in Pithampur, Madhya Pradesh.
This recruitment drive provides an excellent chance to join a growing pharmaceutical manufacturing organization and gain exposure to regulated manufacturing operations, quality systems, and modern pharmaceutical technologies. Gepach Hiring for QA QC FD Production Engineering Roles
Job Overview
| Job Title | QA, QC, F&D, Production, Engineering Professionals |
|---|---|
| Company | Gepach International Pvt. Ltd. |
| Industry | Pharmaceutical Manufacturing |
| Job Location | Pithampur, Dhar, Madhya Pradesh |
| Employment Type | Full-Time |
| Interview Type | Walk-In Interview |
| Experience Required | Freshers to 8 Years |
| Education | B.Sc, M.Sc, B.Pharm, M.Pharm, ITI, Diploma |
| Interview Date | 15 March 2026 |
| Interview Time | 10:00 AM – 5:30 PM |
| Department | Quality, Production, Engineering, Formulation |
| Salary | ₹2.5 LPA – ₹8 LPA (depending on experience) |
Company Overview
Gepach International Pvt. Ltd. is a pharmaceutical manufacturing organization engaged in the development and production of pharmaceutical formulations. The company operates from its manufacturing facility located in the Indore Special Economic Zone (SEZ) at Pithampur, one of India’s important pharmaceutical and industrial hubs.
The organization focuses on maintaining high manufacturing standards and regulatory compliance while delivering quality pharmaceutical products. With its expanding operations, Gepach International continues to recruit talented professionals across multiple departments to strengthen its manufacturing and quality teams.
The company provides a professional working environment where employees can gain experience in regulated pharmaceutical production systems, quality management practices, and modern manufacturing technologies. Gepach Hiring for QA QC FD Production Engineering Roles
Job Location & Employment Type
Work Location:
Pithampur, Dhar District, Madhya Pradesh, India
Employment Type:
Full-Time
Candidates selected through this recruitment drive will be placed at the company’s manufacturing facility located in the Indore Special Economic Zone (SEZ), Phase II.
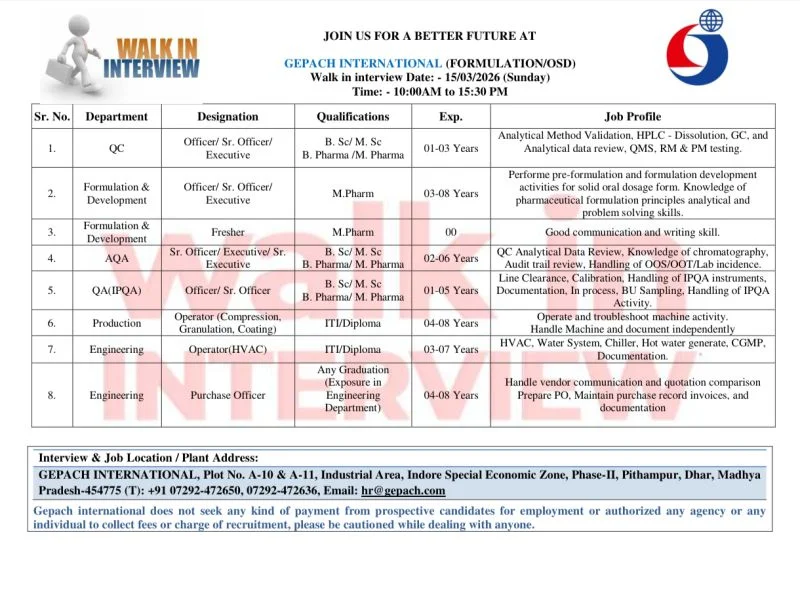
Open Positions / Department Details
Gepach International is recruiting professionals across multiple departments within its pharmaceutical manufacturing operations.
Quality Control
- Officer
- Senior Officer
- Executive
Formulation & Development (F&D)
- Officer
- Senior Officer
- Executive
- Freshers (M.Pharm candidates)
Analytical Quality Assurance (AQA)
- Officer
- Senior Officer
- Executive
Quality Assurance – IPQA
- Officer
- Senior Officer
Production
- Machine Operators for:
- Compression
- Granulation
- Coating
Engineering Department
- HVAC Operator
- Purchase Officer
These roles provide opportunities for candidates interested in quality systems, pharmaceutical formulation development, manufacturing operations, and engineering maintenance. Gepach Hiring for QA QC FD Production Engineering Roles
Key Roles & Responsibilities
Quality Control
Professionals working in Quality Control will be responsible for laboratory testing and analytical activities related to pharmaceutical products. Gepach Hiring for QA QC FD Production Engineering Roles
Main responsibilities include:
- Performing analytical method validation activities
- Operating analytical instruments such as HPLC, GC, and dissolution systems
- Conducting routine analysis of pharmaceutical samples
- Reviewing analytical data and ensuring regulatory compliance
- Supporting quality management system (QMS) documentation
- Maintaining accurate laboratory records and reports
Formulation & Development (F&D)
The Formulation & Development department focuses on developing pharmaceutical formulations and optimizing drug delivery systems. Gepach Hiring for QA QC FD Production Engineering Roles
Key responsibilities include:
- Conducting pre-formulation studies for new products
- Developing solid oral dosage formulations
- Applying pharmaceutical formulation principles in product development
- Troubleshooting formulation and analytical challenges
- Collaborating with analytical and production teams during development stages
Analytical Quality Assurance (AQA)
Analytical QA professionals oversee laboratory compliance and ensure analytical data integrity. Gepach Hiring for QA QC FD Production Engineering Roles
Responsibilities include:
- Reviewing analytical reports generated by QC laboratories
- Managing audit trail reviews
- Investigating laboratory deviations
- Handling OOS (Out of Specification) and OOT (Out of Trend) cases
- Ensuring laboratory documentation meets regulatory standards
- Supporting internal and external audits
Quality Assurance – IPQA
The IPQA team ensures product quality during the manufacturing process.
Primary responsibilities include:
- Performing line clearance before production activities
- Conducting in-process checks during manufacturing
- Monitoring production operations for compliance
- Verifying batch manufacturing documentation
- Managing sampling procedures
- Preparing quality inspection reports
Production Department
Production operators are responsible for running pharmaceutical manufacturing equipment. Gepach Hiring for QA QC FD Production Engineering Roles
Key responsibilities include:
- Operating machines such as compression, granulation, and coating equipment
- Monitoring production parameters
- Maintaining equipment records and production logs
- Following cGMP guidelines during manufacturing
- Supporting batch manufacturing processes
Engineering Department – HVAC
Engineering professionals manage facility systems that support pharmaceutical manufacturing. Gepach Hiring for QA QC FD Production Engineering Roles
Responsibilities include:
- Operating HVAC systems
- Maintaining water systems and chillers
- Ensuring engineering systems comply with cGMP standards
- Conducting preventive maintenance
- Maintaining engineering documentation and records
Purchase Officer
The Purchase Officer manages procurement activities within the engineering or operations department.
Key responsibilities include:
- Coordinating with vendors and suppliers
- Comparing quotations and negotiating procurement terms
- Preparing purchase orders
- Maintaining purchase and invoice records
- Managing procurement documentation
Eligibility Criteria
Educational Qualification
Candidates must have one of the following qualifications:
- B.Sc or M.Sc (Science)
- B.Pharm or M.Pharm (Pharmacy)
- M.Pharm (Freshers eligible for F&D)
- ITI or Diploma (for Production and Engineering roles)
- Any graduate with procurement experience for Purchase Officer role
Experience Required
- Freshers eligible for selected roles (particularly M.Pharm candidates in F&D)
- 1 to 8 years of relevant pharmaceutical industry experience for experienced positions
Gepach Hiring for QA QC FD Production Engineering Roles
Skills Required
Candidates applying for these roles should ideally possess:
- Knowledge of pharmaceutical manufacturing processes
- Understanding of cGMP and regulatory requirements
- Analytical laboratory experience
- Documentation and reporting skills
- Problem-solving ability
- Team collaboration and communication skills
- Familiarity with pharmaceutical equipment and systems
Salary & Benefits
The salary offered will depend on candidate qualifications, experience, and role.
Estimated Salary Range:
₹2.5 LPA – ₹8 LPA
Additional benefits may include:
- Professional development opportunities
- Exposure to regulated pharmaceutical manufacturing systems
- Career growth within the pharmaceutical industry
- Learning environment focused on compliance and quality
Gepach Hiring for QA QC FD Production Engineering Roles
Selection Process
The recruitment process will be conducted through a walk-in interview.
Selection steps may include:
- Document verification
- HR screening interview
- Technical discussion with department heads
- Final evaluation based on qualifications and experience
Candidates should ensure they bring all required documents for verification.
How to Apply
Eligible candidates can attend the walk-in interview directly at the following venue:
Company:
Gepach International Pvt. Ltd.
Interview Address:
Plot No. A-10 & A-11
Industrial Area
Indore Special Economic Zone (SEZ) Phase-II
Pithampur, Dhar
Madhya Pradesh – 454775
Contact Details:
Phone: 091-07292-472650 / 07292-472636
Email: hr@gepach.com
Candidates should carry:
- Updated resume
- Educational qualification certificates
- Experience letters (if applicable)
- Government ID proof
- Passport size photographs
Important Dates
| Event | Date |
|---|---|
| Walk-In Interview Date | 15 March 2026 |
| Interview Time | 10:00 AM – 5:30 PM |
| Job Location | Pithampur, Dhar, Madhya Pradesh |
Why Apply for This Pharma Job?
Joining Gepach International offers several advantages for professionals seeking long-term careers in the pharmaceutical industry.
Key benefits include:
- Opportunity to work in a regulated pharmaceutical manufacturing environment
- Exposure to advanced production technologies and quality systems
- Career opportunities across multiple departments
- Professional growth and learning opportunities
- Experience in pharmaceutical development, manufacturing, and quality assurance processes
This recruitment drive is particularly beneficial for fresh graduates as well as professionals with experience in pharma manufacturing operations. Gepach Hiring for QA QC FD Production Engineering Roles
Important Note / Disclaimer
IndiaPharmaJobs.in is an independent job information platform that publishes pharmaceutical industry job updates for informational purposes only. We are not affiliated with Gepach International or any recruitment agency.
Candidates should apply or attend interviews directly through official company channels. The company does not charge any recruitment or placement fees, and applicants should be cautious of fraudulent job offers. Gepach Hiring for QA QC FD Production Engineering Roles
Final Call-to-Action
If you are a qualified candidate looking to advance your career in the pharmaceutical industry, this walk-in opportunity at Gepach International could be the next step in your professional journey. Candidates with relevant qualifications and experience are encouraged to attend the interview with all necessary documents and explore career opportunities within a growing pharmaceutical manufacturing organization. Gepach Hiring for QA QC FD Production Engineering Roles

