Regulatory Affairs Specialist Fresher Job at Saraca Solutions in Noida for biotechnology graduates interested in medical device compliance and regulatory documentation.
The medical device and healthcare technology sector is experiencing significant growth worldwide, leading to increased demand for professionals skilled in regulatory affairs, compliance documentation, and quality management systems. Companies involved in medical device development must comply with global regulatory frameworks such as USFDA guidelines, European Union Medical Device Regulation (EU MDR), and Indian regulatory standards.
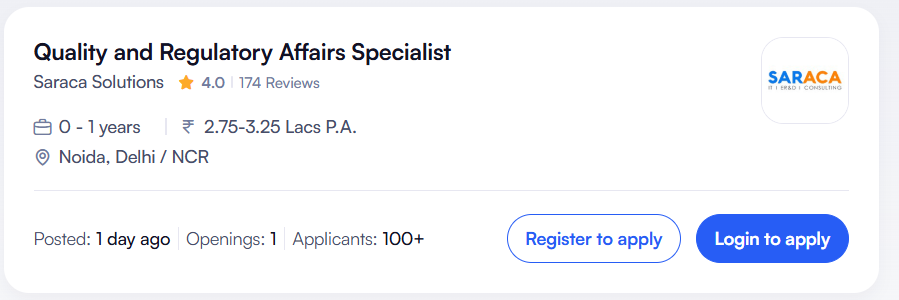
Saraca Solutions has announced an excellent career opportunity for fresh graduates interested in Regulatory Affairs and Quality Compliance. The company is hiring for the position of Regulatory Affairs Specialist in Noida, Delhi NCR.
This position offers an ideal starting point for candidates who want to build a career in medical device regulatory affairs, technical documentation, and global compliance management. Fresh graduates from biotechnology and life sciences backgrounds are encouraged to apply for this entry-level regulatory role. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Company Overview
Saraca Solutions is a technology-driven engineering and consulting organization that provides specialized services in product engineering, regulatory compliance, and medical device consulting. The company works with healthcare and life sciences organizations to help them meet global regulatory requirements and maintain high-quality standards in product development.
Saraca Solutions supports companies operating in international markets by assisting with regulatory documentation, quality management systems, and compliance frameworks required for medical device approvals. Through its regulatory consulting expertise, the company contributes to ensuring that medical technologies meet safety and performance standards worldwide.
Professionals working with Saraca Solutions gain valuable exposure to international medical device regulations, compliance systems, and technical documentation practices used across global healthcare markets. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Job Overview (Job Details)
| Job Title | Regulatory Affairs Specialist |
|---|---|
| Company | Saraca Solutions |
| Department | Research & Development / Regulatory Affairs |
| Job Location | Noida, Delhi NCR, India |
| Employment Type | Full-Time, Permanent |
| Experience Required | 0 – 1 Years (Freshers Eligible) |
| Qualification | B.Tech Biotechnology or related Life Sciences degree |
| Industry | Medical Devices / Biotechnology |
| Salary Range | ₹2.75 – ₹3.25 LPA (Approx.) |
| Application Mode | Online Application |
Job Location & Employment Type
- Location: Noida, Delhi NCR, India
- Employment Type: Full-Time Permanent
- Department: Research & Development / Regulatory Affairs
This position involves supporting regulatory compliance activities for medical devices intended for global markets including the United States, Europe, and India. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Open Position
Regulatory Affairs Specialist (Quality & Regulatory Affairs)
The selected candidate will assist in regulatory documentation, compliance processes, and quality management activities related to medical device products. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Key Roles & Responsibilities
Professionals selected for this position will contribute to various regulatory and quality compliance activities within the organization.
Regulatory Documentation
- Prepare and maintain regulatory documentation related to medical device products.
- Assist in compiling technical files required for regulatory submissions.
- Ensure documentation complies with applicable regulatory standards.
Global Regulatory Compliance
- Support compliance activities related to USFDA regulations, EU MDR requirements, and Indian medical device regulations.
- Monitor regulatory guidelines and ensure documentation aligns with updated compliance requirements.
Technical Documentation Support
- Assist in preparing technical documents required for regulatory approvals.
- Maintain documentation systems for regulatory submissions and approvals.
Medical Writing Activities
- Contribute to preparation of medical writing documents and clinical evaluation reports.
- Ensure documentation meets regulatory standards and submission requirements.
Product Documentation Review
- Review product-related documents to ensure they comply with regulatory guidelines.
- Verify accuracy and completeness of regulatory files.
Quality Management System Support
- Assist in maintaining Quality Management System (QMS) documentation.
- Support quality assurance processes related to regulatory compliance.
Cross-Functional Collaboration
- Coordinate with internal teams to ensure regulatory requirements are incorporated into product documentation.
- Provide regulatory compliance support during product development stages.
Regulatory Monitoring
- Track updates in medical device regulations and compliance requirements.
- Assist in implementing regulatory updates within company documentation processes.
Eligibility Criteria
Candidates applying for this regulatory affairs position should meet the following requirements.
Education
Applicants must possess:
- B.Tech in Biotechnology
or
- A related Life Sciences degree relevant to biotechnology, biomedical sciences, or healthcare technology.
Fresh graduates are eligible to apply for this entry-level role.
Experience
- 0 – 1 years of experience
- Freshers with strong interest in regulatory affairs, medical devices, or compliance documentation are encouraged to apply.
Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Skills Required
Candidates should have the following knowledge and professional skills:
- Basic understanding of Regulatory Affairs in medical devices
- Knowledge of EU MDR (European Union Medical Device Regulation)
- Familiarity with USFDA medical device guidelines
- Understanding of quality documentation and compliance procedures
- Ability to prepare technical documentation and regulatory reports
- Interest in medical writing and clinical documentation
- Good communication and documentation skills
- Attention to detail and analytical thinking
Candidates who have knowledge of international regulatory frameworks will have an additional advantage. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Salary & Benefits
Saraca Solutions offers a competitive entry-level salary package.
Expected Salary Range:
₹2.75 LPA – ₹3.25 LPA (approximately)
Additional benefits may include:
- Exposure to global regulatory systems
- Experience with international compliance frameworks
- Training and development opportunities in regulatory affairs
- Career growth in medical device regulatory and compliance domains
Selection Process
The recruitment process may include the following stages:
- Application screening
- Technical interview focusing on regulatory knowledge
- HR discussion
- Final selection
Only shortlisted candidates will be contacted for further recruitment steps. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
How to Apply
Interested candidates should apply through the official application link provided by the company.
Applicants should prepare an updated resume highlighting:
- Biotechnology or life sciences education
- Knowledge of regulatory affairs
- Interest in medical device compliance
- Technical documentation or research experience
Important Dates
- Application Deadline: Not specified
Candidates are advised to apply early as positions may close once suitable candidates are selected. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Why Apply for This Pharma Job?
This role offers an excellent entry point into the medical device regulatory affairs industry, which is one of the fastest-growing sectors in healthcare. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Key advantages include:
- Opportunity to start a career in medical device regulatory affairs
- Exposure to global compliance frameworks including EU MDR and USFDA
- Hands-on experience in technical documentation and regulatory submissions
- Skill development in medical writing and quality management systems
- Strong foundation for future careers in regulatory affairs, quality assurance, and compliance
For fresh graduates interested in healthcare regulations and medical device compliance, this role provides valuable industry exposure. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Important Note
IndiaPharmaJobs.in is an independent job information platform that publishes pharmaceutical and healthcare job updates for informational purposes only.
We are not affiliated with any company mentioned in this job post and do not charge any fee for job applications. Candidates should always apply through official company recruitment channels. Regulatory Affairs Specialist Fresher Job at Saraca Solutions
Final Call-to-Action
Fresh graduates with a biotechnology or life sciences background who are interested in regulatory affairs and medical device compliance should consider applying for this opportunity. This role can help you build a strong foundation in regulatory affairs and open doors to global career opportunities in the medical device industry. Regulatory Affairs Specialist Fresher Job at Saraca Solutions

