Study Review Specialist Job at ICON plc
Apply for Study Review Specialist job at ICON plc 2026. Check eligibility, clinical research experience, and application process details now online.
Introduction
The clinical research industry is increasingly focusing on data accuracy, regulatory compliance, and efficient document management. The Study Review Specialist Job at ICON plc offers an excellent opportunity for professionals with experience in clinical documentation and study start-up activities.
This role is ideal for candidates who want to work in a global CRO environment, ensuring high-quality clinical trial documentation while collaborating with international teams.
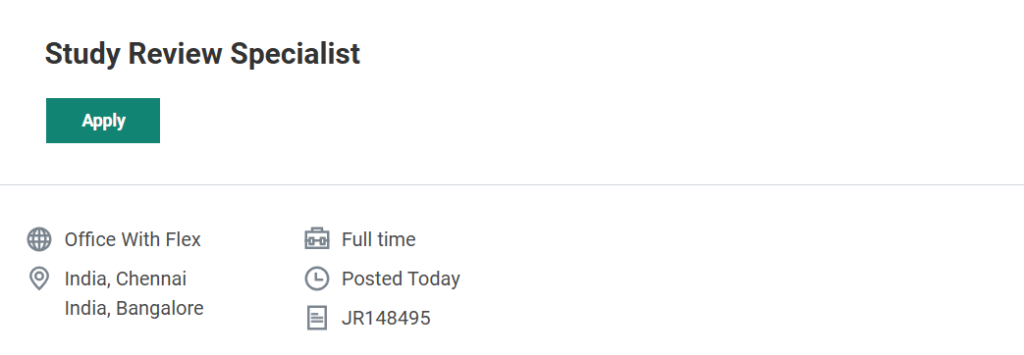
Job Overview
| Particulars | Details |
|---|---|
| Job Title | Study Review Specialist |
| Company Name | ICON plc |
| Location | Chennai / Bangalore, India |
| Employment Type | Full-Time |
| Department | Clinical Research / Document Review |
| Experience Required | 1–3 Years |
| Education | Bachelor’s in Life Sciences or related field |
| Salary | As per company norms |
Company Overview
ICON plc is a world-leading healthcare intelligence and clinical research organization. The company provides outsourced development and commercialization services to pharmaceutical, biotechnology, and medical device industries.
ICON is known for its strong global presence, innovative clinical solutions, and commitment to improving patient outcomes through high-quality research and data integrity.
Job Location & Employment Type
This is a full-time position with an “Office with Flex” work model based in Chennai or Bangalore. Candidates may have hybrid flexibility depending on project requirements.
Open Positions / Department Details
The role is part of the Clinical Document Review and Study Start-up team, responsible for reviewing study documents, ensuring compliance, and maintaining quality standards across clinical trials.
Key Roles & Responsibilities
- Review and approve Critical Document Packages (CDPs) before investigational product release
- Ensure compliance with SOPs, ICH/GCP guidelines, and regulatory requirements
- Maintain accurate records of document review and approval activities
- Monitor timelines and proactively escalate delays in document approvals
- Conduct internal quality checks on Trial Master File (TMF) documents
- Collaborate with cross-functional teams to ensure smooth study execution
- Act as a Subject Matter Expert (SME) for assigned processes
- Participate in study meetings and support communication across teams
- Mentor new team members and provide training support
- Contribute to process improvements and technology adoption initiatives
Eligibility Criteria
Education
- Bachelor’s degree in Life Sciences or related discipline
Experience
- 1–3 years of experience in clinical research
- Hands-on experience in study start-up document review is mandatory
- Experience in Contract QC or document quality review is preferred
Skills Required
- Strong understanding of clinical trial processes
- Knowledge of ICH/GCP guidelines and regulatory requirements
- Experience in document management systems and tracking tools
- High attention to detail and accuracy
- Strong communication and collaboration skills
- Ability to manage timelines and multiple tasks
Salary & Benefits
- Salary: As per company norms
- Competitive compensation package
- Health insurance benefits for employees and families
- Retirement planning and savings options
- Work-life balance initiatives and flexible work model
- Employee Assistance Program (24/7 support)
- Life insurance coverage
- Additional benefits like gym memberships, childcare support, and travel benefits (country-specific)
Selection Process
The hiring process generally includes:
- Application screening
- Technical/functional interview (clinical research & document review)
- HR discussion
- Final selection
How to Apply
Candidates can apply through the official online application portal of the company. Ensure your resume highlights your experience in clinical documentation, study start-up, and regulatory compliance.
Important Dates
- Last Date to Apply: Not specified (Apply early for better chances)
Why Apply for This Job?
The Study Review Specialist Job at ICON plc is a valuable opportunity for professionals looking to build expertise in clinical documentation and regulatory compliance.
Here’s why this role stands out:
- Work with a globally recognized CRO
- Gain hands-on experience in clinical trial documentation
- Exposure to international regulatory standards
- Opportunity to grow into regulatory affairs or clinical operations roles
- Flexible work model supporting work-life balance
This position is particularly suitable for candidates aiming to advance in clinical research, regulatory affairs, or quality assurance domains.
FAQs
1. What is the eligibility for Study Review Specialist Job at ICON plc?
Candidates must have a bachelor’s degree in Life Sciences with 1–3 years of clinical research experience.
2. Is study start-up experience mandatory?
Yes, hands-on experience in reviewing study start-up documents is a primary requirement.
3. What is the work location?
The job is based in Chennai or Bangalore with flexible office options.
4. What skills are required for this role?
Knowledge of ICH/GCP, document review, and clinical trial processes is essential.
5. Is this role suitable for freshers?
No, candidates need at least 1 year of relevant experience.
Conclusion
The Study Review Specialist Job at ICON plc 2026 offers an excellent platform for professionals to build a career in clinical documentation and regulatory compliance. With global exposure, strong benefits, and growth opportunities, this role is ideal for candidates looking to advance in the clinical research field.
Interested candidates should apply as soon as possible to maximize their chances.
Disclaimer
IndiaPharmaJobs.in is an independent job information platform. We do not provide jobs or recruitment and are not affiliated with any company.

