Technical Writer I Job in Bangalore Thermo Fisher Scientific. Bioscience technical writing role. Apply online before 31 March 2026.
Professionals with a strong background in biosciences and technical documentation now have an excellent opportunity in Bangalore. A full-time, onsite role is open for Technical Writer I, offering the chance to contribute to high-impact scientific documentation within a globally recognized organization. Candidates who enjoy transforming complex scientific data into clear, structured, and user-friendly content can build a rewarding career in this dynamic environment. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Company Overview
Thermo Fisher Scientific is a globally respected biotechnology and life sciences company dedicated to enabling customers to make the world healthier, cleaner, and safer. The organization supports scientific research, healthcare innovation, and laboratory advancements through cutting-edge technologies and services. With a strong presence in India, it provides professionals with opportunities to work on globally impactful projects while collaborating with cross-functional international teams. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Job Location & Employment Type
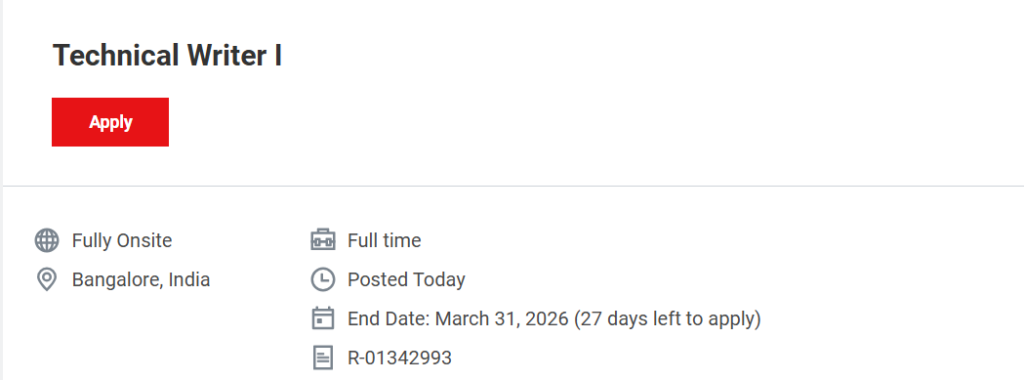
- Location: Bangalore, Karnataka, India
- Work Mode: Fully Onsite
- Employment Type: Full-Time
- Work Schedule: Standard (Monday–Friday)
- Work Environment: Office
Position Details
Job Title: Technical Writer I
Job Requisition ID: R-01342993
Application Deadline: 31 March 2026
This role focuses on creating and maintaining high-quality technical documentation for bioscience products, ensuring accuracy, regulatory alignment, and clarity for end users. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Department Overview
The Technical Documentation function plays a vital role in supporting product development and customer enablement. The team works closely with research & development, product management, quality assurance, and regulatory affairs departments to ensure all documentation meets internal standards and industry expectations. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Key Roles & Responsibilities
The selected candidate will contribute to the documentation lifecycle by performing the following duties:
- Develop and maintain user manuals, product guides, and technical documentation for bioscience products
- Collaborate with subject matter experts, engineers, and project managers to gather technical information
- Translate complex scientific data into clear, concise, and structured documentation
- Ensure alignment of documents with company standards, business unit guidelines, and regulatory expectations
- Review and update existing materials to improve clarity, usability, and consistency
- Manage multiple documentation projects simultaneously while adhering to deadlines
- Maintain organized documentation repositories and reusable templates
- Support documentation quality improvements through structured content management practices
- Participate in required training programs and stay updated with technical and scientific developments
This role demands precision, structured thinking, and the ability to work both independently and collaboratively. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Eligibility Criteria
Education
- Bachelor’s degree in Biosciences or a related discipline
- Advanced degree in Biosciences is preferred
Equivalent experience in a relevant scientific field may also be considered.
Experience
- Prior experience in technical writing, preferably within bioscience or life sciences sectors
- Experience in preparing scientific documentation, manuals, or product-related materials
- Exposure to structured authoring environments is advantageous
Skills Required
- Strong written and verbal communication skills
- Ability to simplify and present complex scientific information clearly
- High level of attention to detail and documentation accuracy
- Familiarity with industry terminology and documentation standards
- Experience with XML/DITA-based authoring tools such as Oxygen XML Editor/Author
- Exposure to component content management systems like RWS Tridion Docs or similar platforms
- Proficiency in Microsoft Office Suite and Adobe Creative tools
- Strong organizational and time management abilities
- Capability to manage multiple assignments under tight timelines
- Willingness to continuously enhance technical knowledge
Candidates with certification in technical writing may have an added advantage.
Salary & Benefits
Compensation and benefits will be offered as per company norms and in accordance with candidate qualifications and experience.
Selection Process
The recruitment process may include:
- Application screening
- Technical writing assessment or assignment
- Interview rounds with technical and hiring managers
- Final selection and offer
Only shortlisted candidates will be contacted for further evaluation.
How to Apply
Interested applicants must submit their application through the official online portal before 31 March 2026. Ensure your resume highlights relevant technical writing experience, scientific background, and familiarity with structured documentation tools.
Early applications are encouraged due to competitive selection. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Important Dates
- Last Date to Apply: 31 March 2026
Candidates should complete their online application well before the deadline.
Why Apply for This Pharma & Life Sciences Job?
Technical writing in the bioscience domain is a high-value skill that bridges research, regulatory, and customer communication. This opportunity offers:
- Exposure to global bioscience product documentation
- Hands-on experience with structured authoring and content management systems
- Opportunity to collaborate with R&D and regulatory experts
- Career growth in scientific documentation and content strategy
- Stability and long-term prospects in a global life sciences organization
Professionals seeking a career that combines scientific knowledge with communication expertise will find this role highly rewarding. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Important Disclaimer
IndiaPharmaJobs.in is an independent job information platform providing pharmaceutical, biotechnology, and healthcare job updates for informational purposes only. We are not affiliated with Thermo Fisher Scientific or any other organization mentioned above. Applicants are advised to verify complete job details through the official company website before applying. Technical Writer I Job in Bangalore Thermo Fisher Scientific
Eligible candidates interested in advancing their career in bioscience technical documentation should apply online before the deadline and take the next step toward building expertise in global scientific communication. Technical Writer I Job in Bangalore Thermo Fisher Scientific

